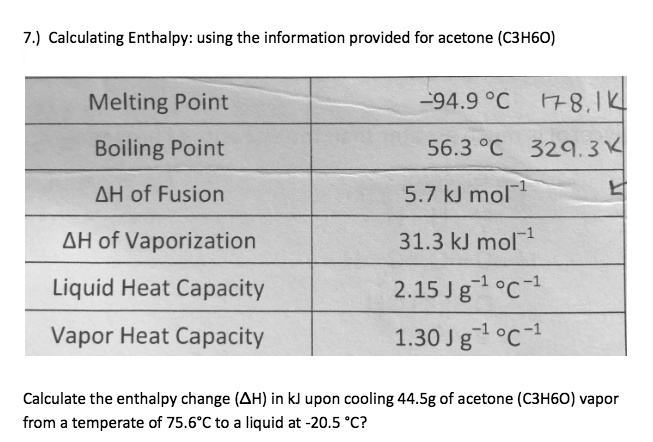
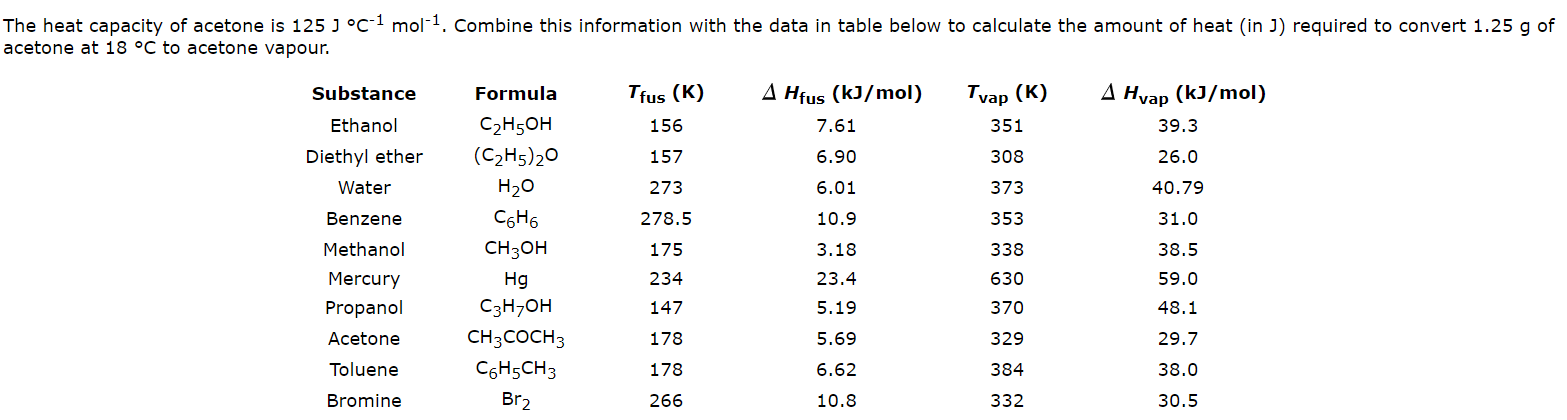
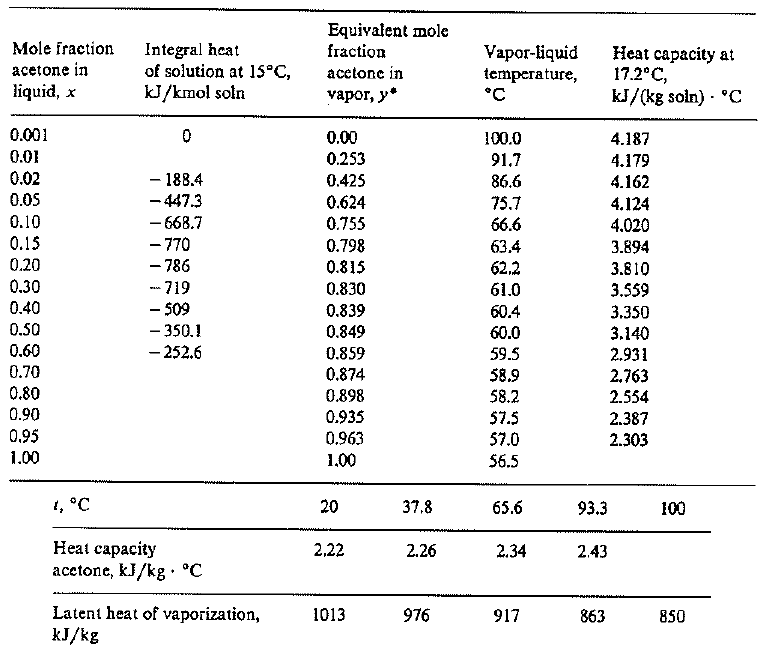
SOLVED: A solid substance has a mass 0f 250.00g It is cooled by 25.00*C and loses 4.937kJ of heat What is the specific heat capacity of the substance in 9.PC [APP 2
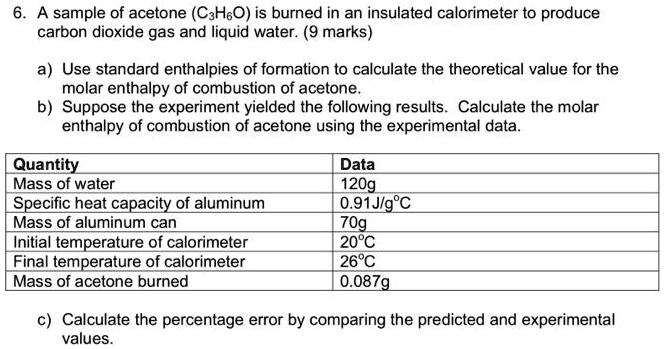
SOLVED: A sample of acetone (CaHsO) is burned in an insulated calorimeter t0 produce carbon dioxide gas and liquid water. (9 marks) Use standard enthalpies of formation to calculate the theoretical value

Using the given data and calculate the enthalpy of formation of acetone(g). Bond enthalpy of : C - H = 415 ; C - C = 350 ; (C = O) =

Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram
THE HEAT CAPACITIES OF ISOPROPYL ALCOHOL AND ACETONE FROM 16 TO 298°K. AND THE CORRESPONDING ENTROPIES AND FREE ENERGIES | Journal of the American Chemical Society
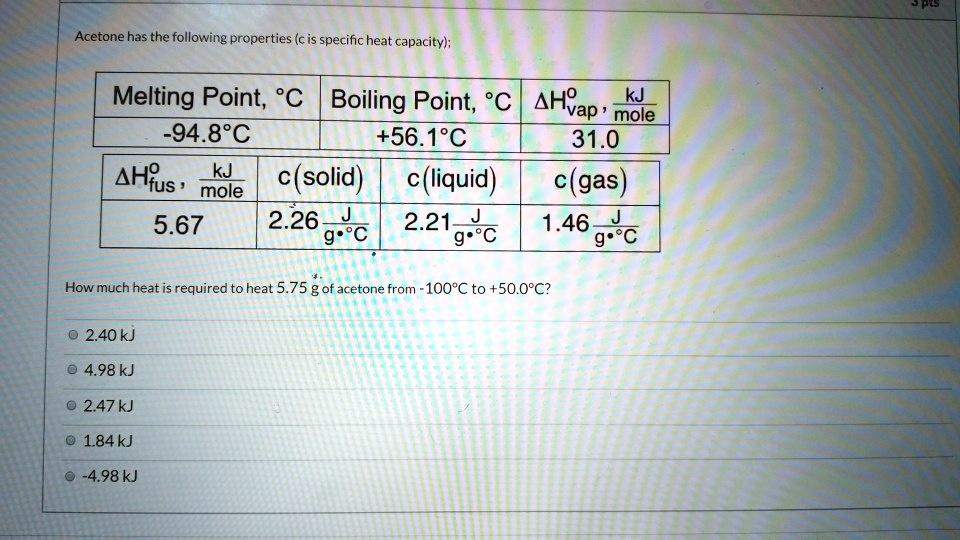
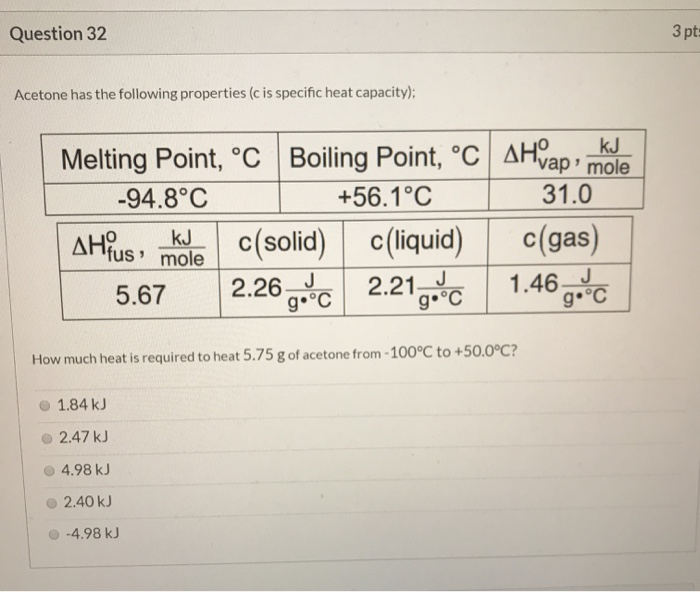
SOLVED: Acetone has the following properties (c is specific heat capacity); Melting Point; %C Boiling Point; C AHYap' kJ mole -94.8'C +56.1*C 31.0 AH?s , kJ mole (solid) (liquid) c(gas) 5.67 2.26-