
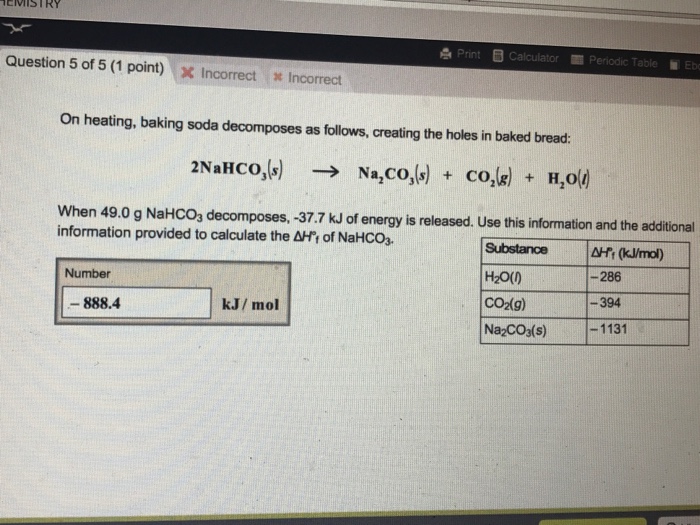
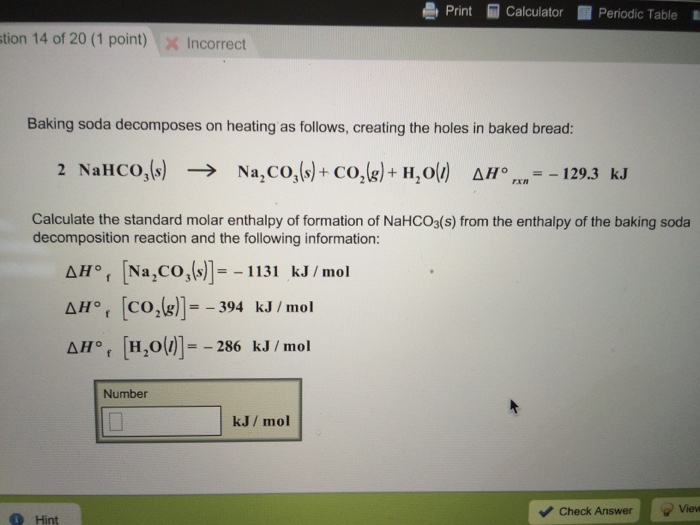
Upon heating, baking soda (sodium hydrogen carbonate) decomposes according to the equation: 2NaHCO(3)(s) to Na(2)CO(3)(s) + H(2)O(l) + CO(2)(g) In an experiment, 8.4 g baking soda decomposes and carbon dioxide gas is

SOLVED: A recipe for scones asks for 3.0 g of baking soda (sodium hydrogen carbonate) . Upon heating at 180 OC at a pressure of 1.00 bar; baking soda decomposes as follows:







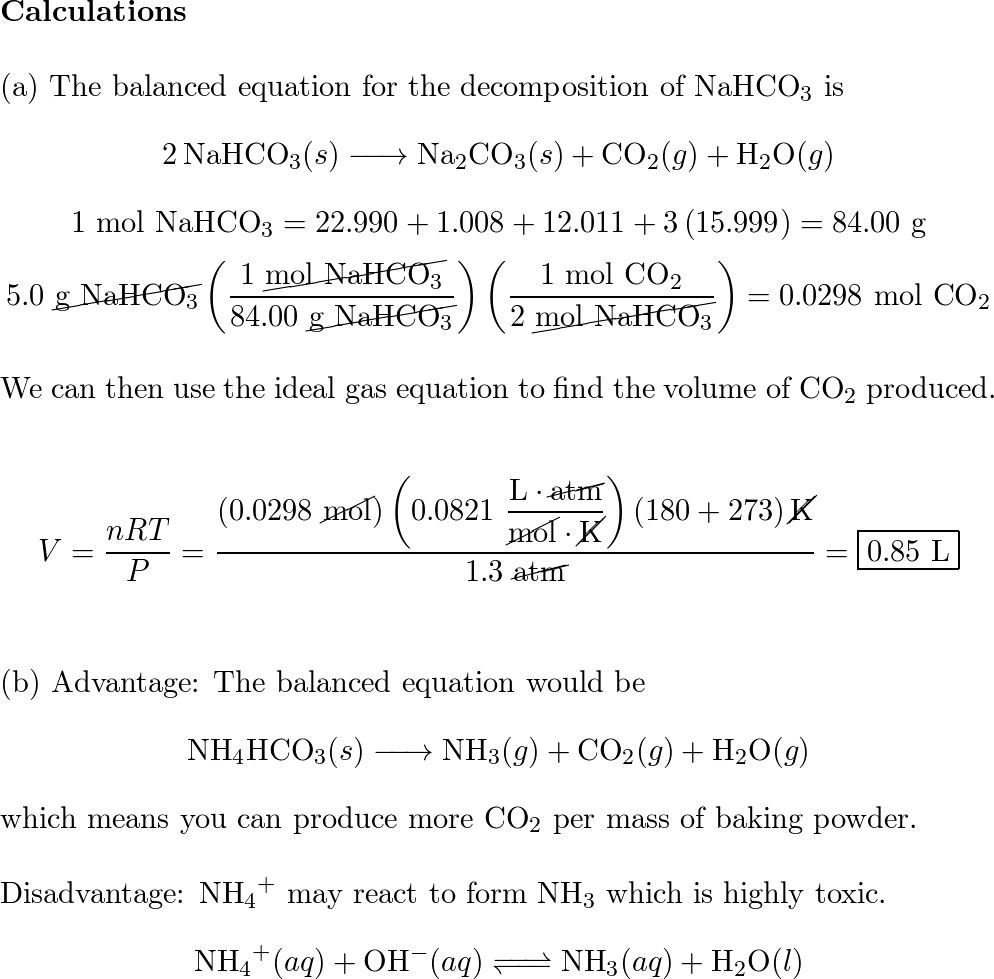







:max_bytes(150000):strip_icc()/185329704-56a1300e5f9b58b7d0bce3ce.jpg)



