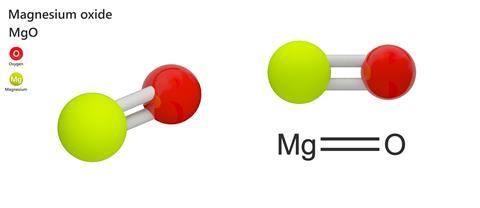![Synthesis of hybrid magnesium hydroxide/magnesium oxide nanorods [Mg(OH)2/MgO] for prompt and efficient adsorption of ciprofloxacin from aqueous solutions - ScienceDirect Synthesis of hybrid magnesium hydroxide/magnesium oxide nanorods [Mg(OH)2/MgO] for prompt and efficient adsorption of ciprofloxacin from aqueous solutions - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0959652622005868-ga1.jpg)
Synthesis of hybrid magnesium hydroxide/magnesium oxide nanorods [Mg(OH)2/MgO] for prompt and efficient adsorption of ciprofloxacin from aqueous solutions - ScienceDirect
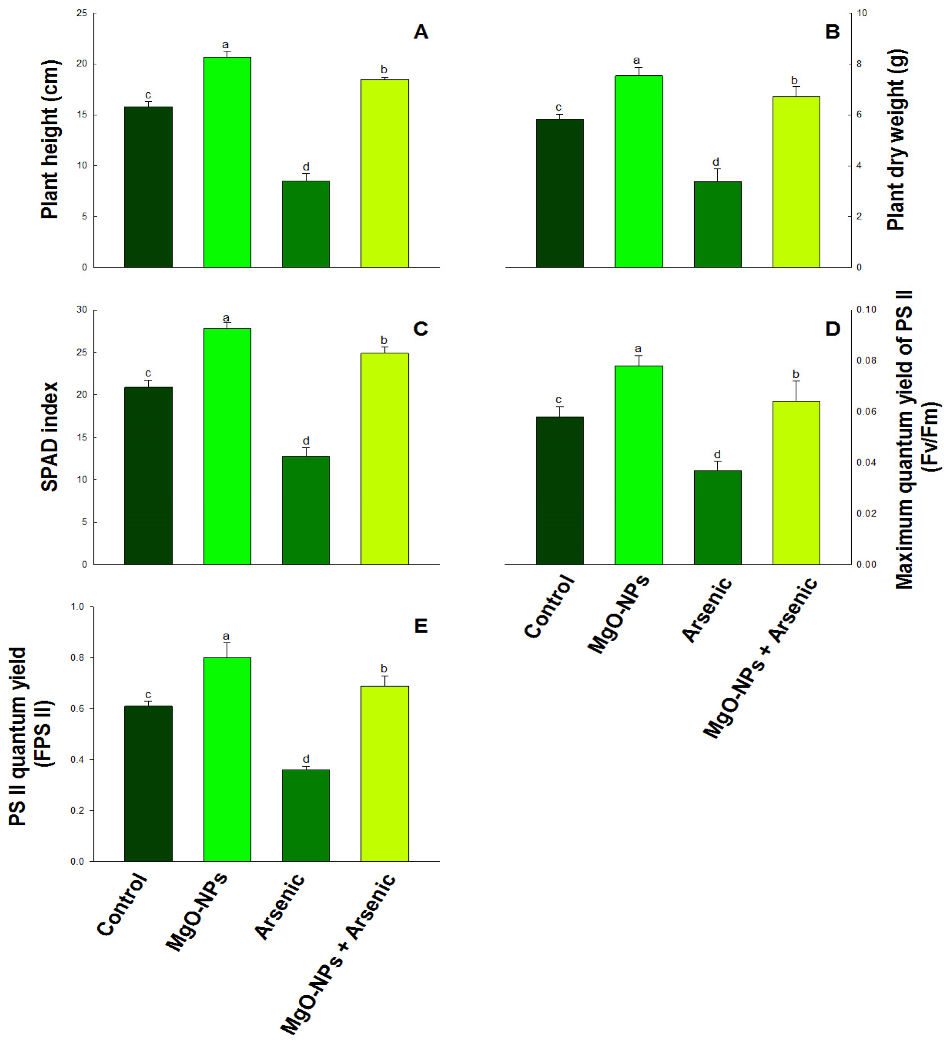
Metals | Free Full-Text | Magnesium Oxide Nanoparticles (MgO-NPs) Alleviate Arsenic Toxicity in Soybean by Modulating Photosynthetic Function, Nutrient Uptake and Antioxidant Potential
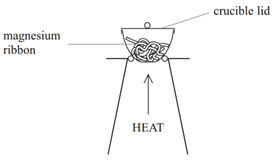
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry
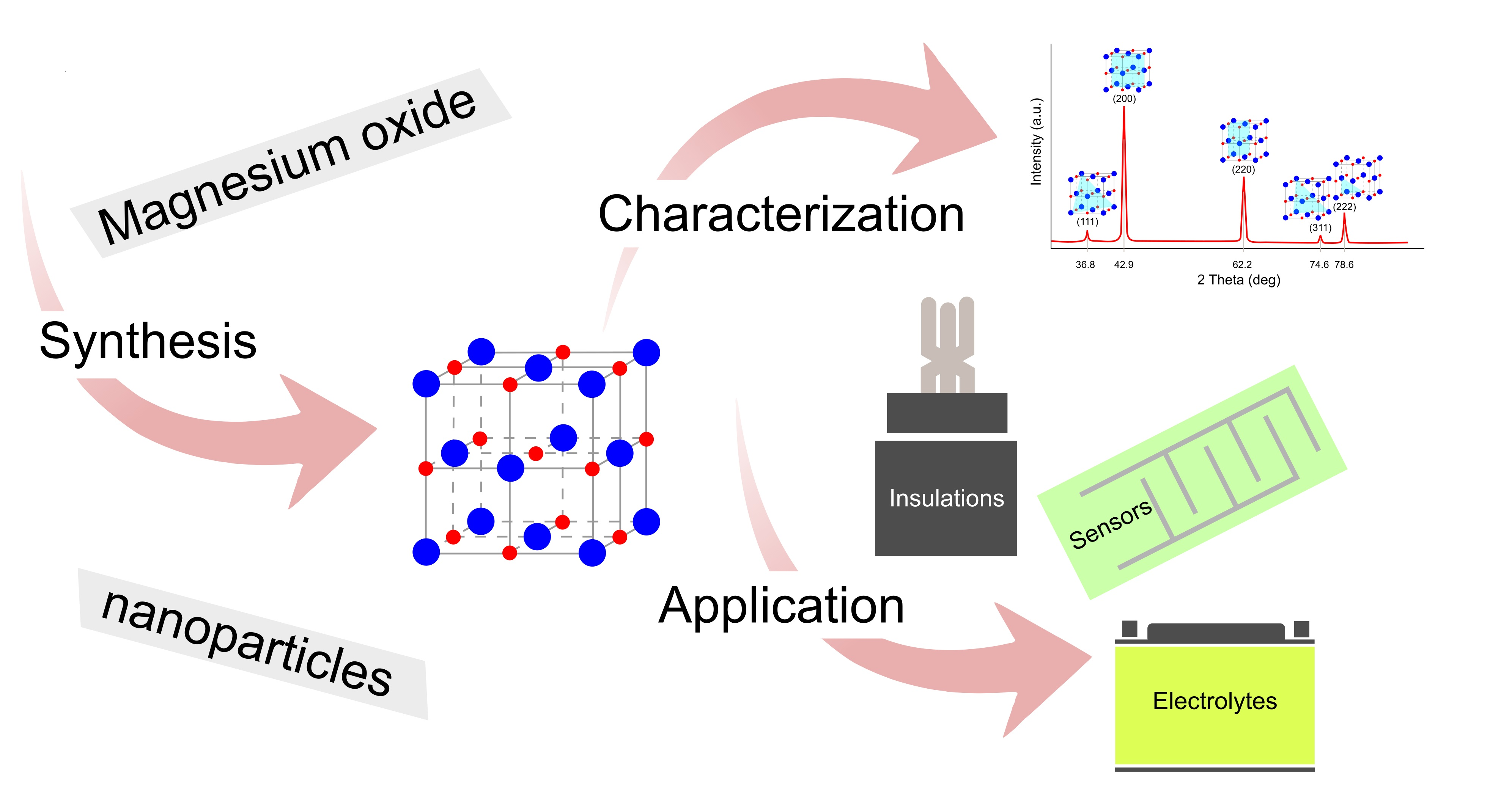
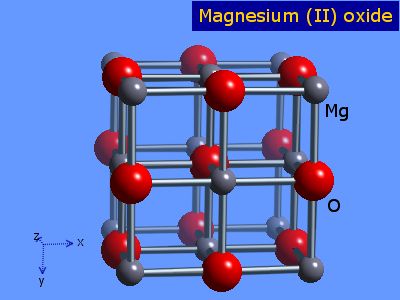
IJMS | Free Full-Text | Synthesis, Properties, and Selected Technical Applications of Magnesium Oxide Nanoparticles: A Review

H 2 SO 4 + Zn 1) Sulphuric acid + zinc 3) Nitric acid + sodium thiosulphate 2) Hydrochloric acid + magnesium 4) Hydrogen peroxide with catalyst Popular. - ppt download

Generally, metallic oxides are basic and non - metallic oxides are acidic in nature. Solution of which of the following oxides in water will change the colour of blue litmus to red?
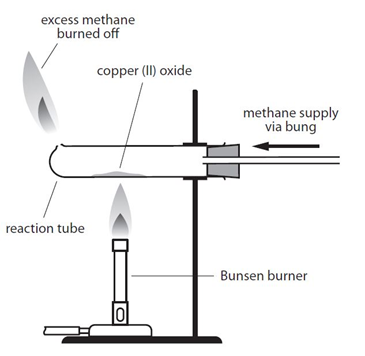
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry
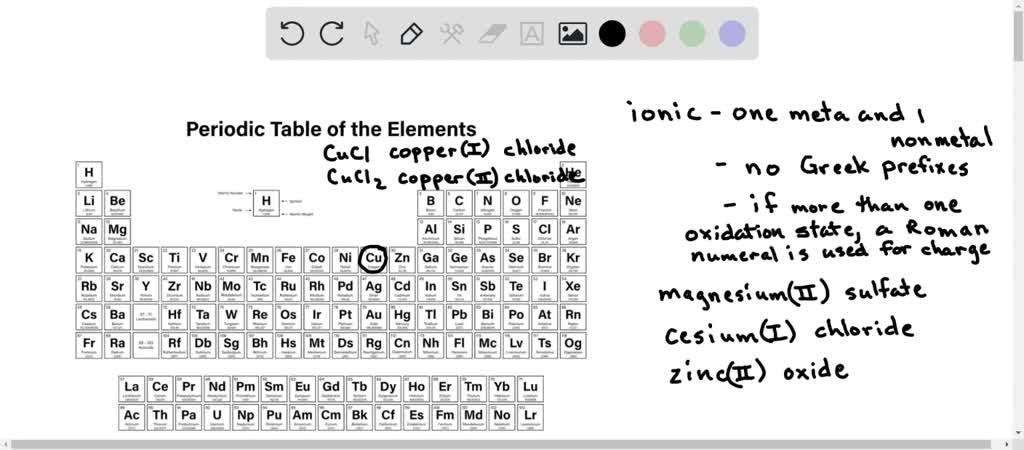
SOLVED: Why are the names magnesium (II) sulfate, cesium (I) chloride, and zinc (II) oxide incorrect? Explain why