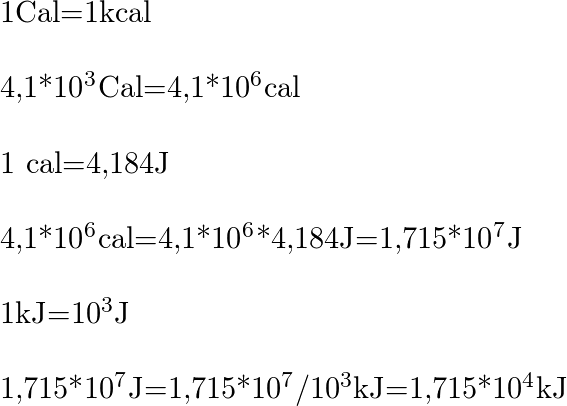A calorie is a unit of heat or energy and it equals about 4.2 J where 1J = 1 kg m2s–2. Suppose we - Brainly.in
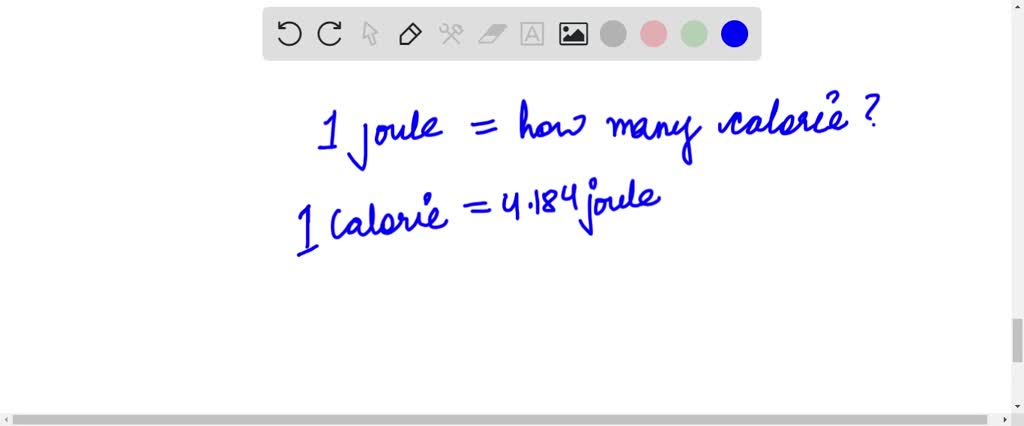
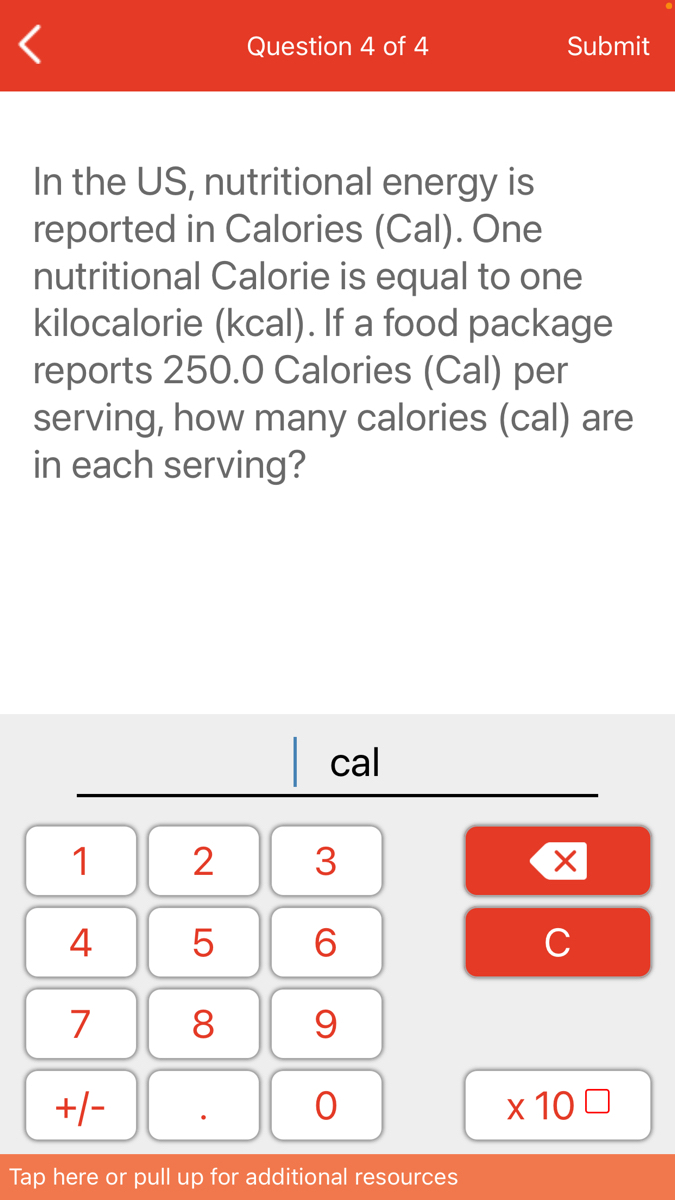
![SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ] SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ]](https://cdn.numerade.com/ask_images/f3f65c4b70424578aa674a37eabc4800.jpg)
SOLVED: One calorie is equal to 4.184J. How much energy in joules is supplied by a breakfast bar containing 100 nutritional calories? Select all the correct answers (2 Points) 420.0 Ki 42 x 10*5 ) 421 17 *1045 ]

Not all calories are equal – a dietitian explains the different ways the kinds of foods you eat matter to your body
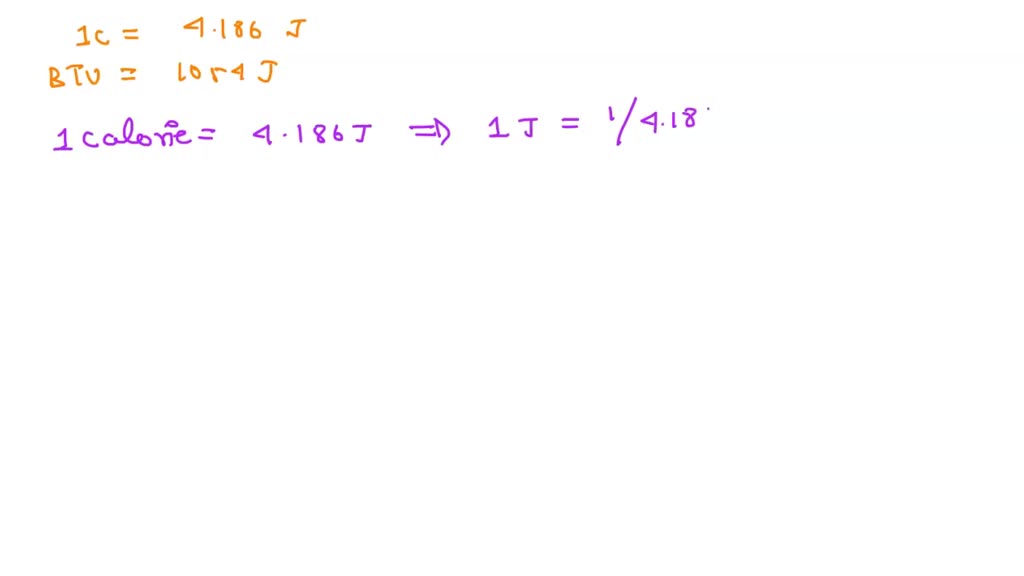
SOLVED: How many calories are equal to one BTU? (One calorie = 4.186 J, one BTU = 1 054 J.) a. 0.252 b. 3.97 c. 252 d. 397
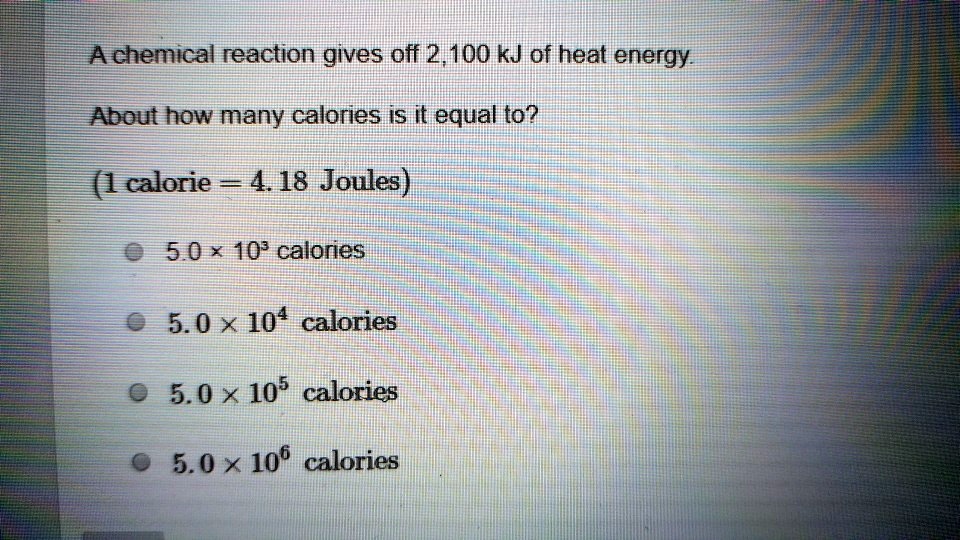
SOLVED: Achemical reaction gives off 2,100 kJ of heat energy About how many calories is it equal to? (1 calorie 4.18 Joules) 8 5.0 * 10: calories 5.0 X 104 calories 5.0 > 105 calories 5.0 > 106 calories
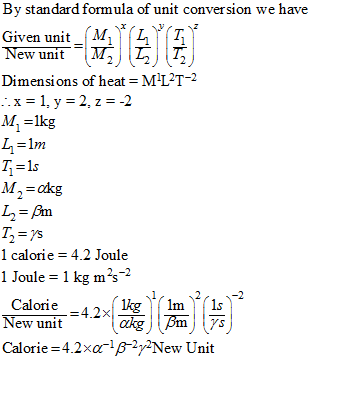
A calorie is a unit of heat or energy and it equals about 4 2 J where 1J = 1 - Physics - Units And Measurements - 2701112 | Meritnation.com

PPT - One watt is equivalent to: one joule/sec one coulomb/sec one calorie/sec one horsepower PowerPoint Presentation - ID:4721076