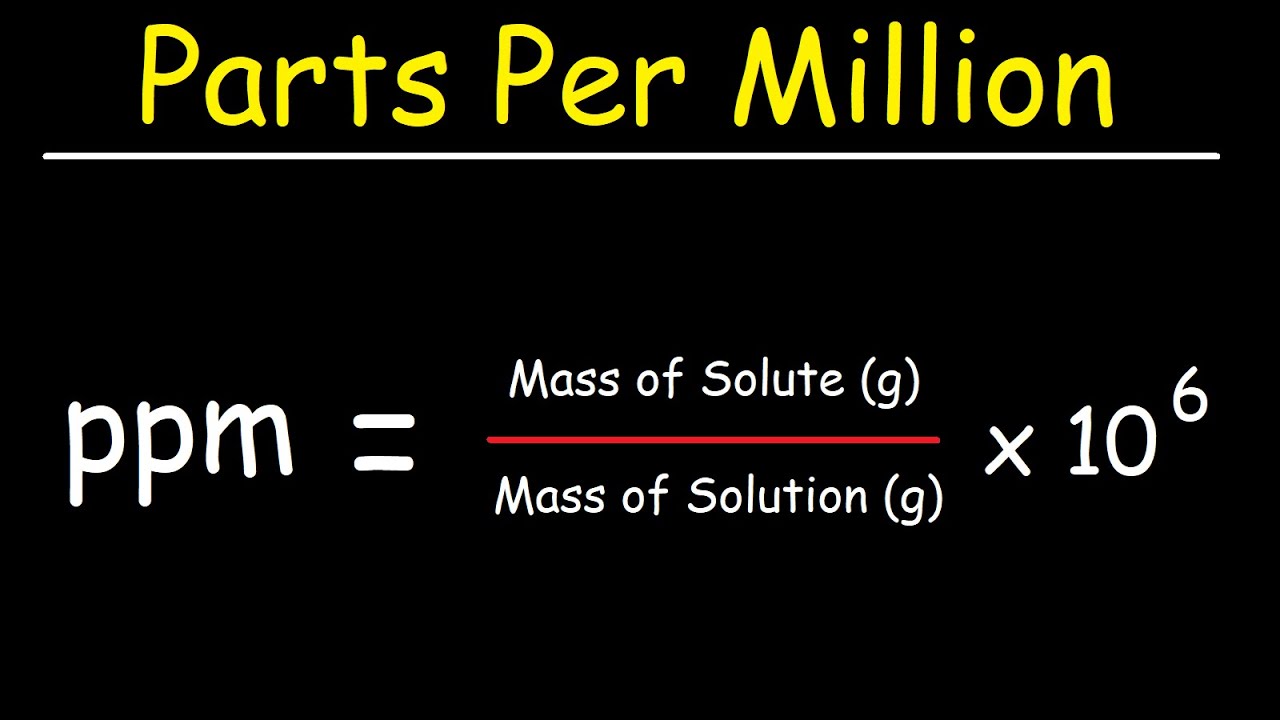SOLVED: the critical concentration of micronutrients needed in tissue is equal to or less than (1) 1 ppm (2) 10 ppm (3) 100 ppm (4) 1000 ppm

Parts per Million Concentration. Parts per Million (ppm) is the ratio of the number of grams of solute for every one million grams of solution. parts. - ppt download

Suppose a pure Si-crystal has 6xx10^(28) "atoms" m^(-3). It is doped by 1 ppm concentration of pentavalent As. Calculate the number of electrons and holes. Give that n(i)=1.5xx10^(16)m^(-3).

A sample of drinking water was found to be severely contaminated with chloroform (CHCl3) supposed to be a carcinogen. The level of contamination was 15 ppm (by mass):(i) express this in percent

10.30 mg of O2 is dissolved into a liter of sea water of density 1.03 g/mL . The concentration of O2 in ppm is .

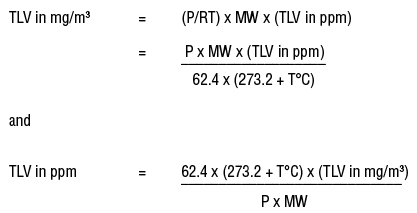
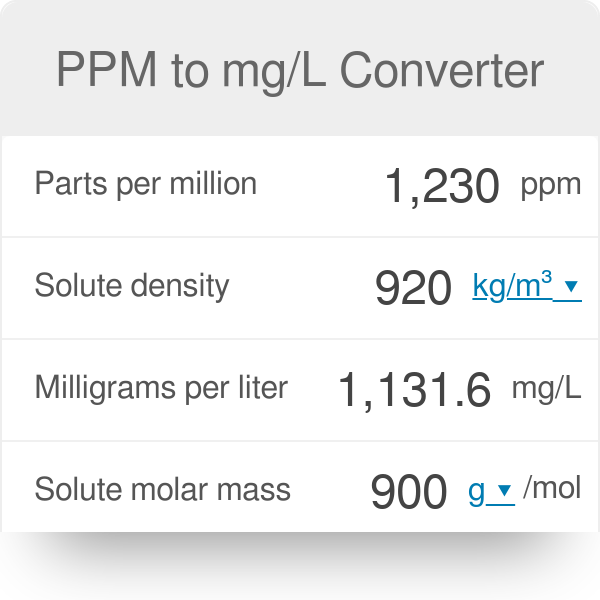
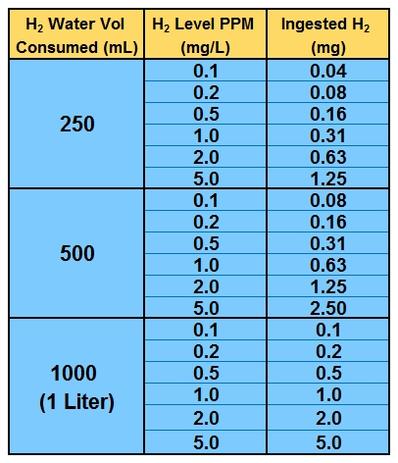
Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download