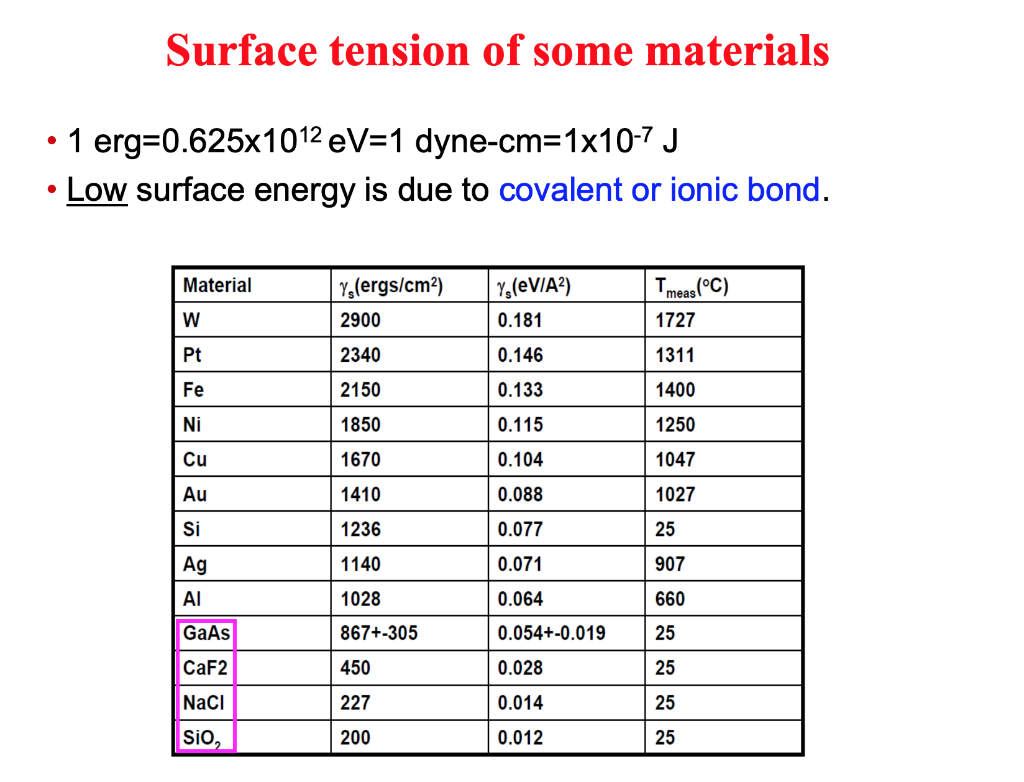
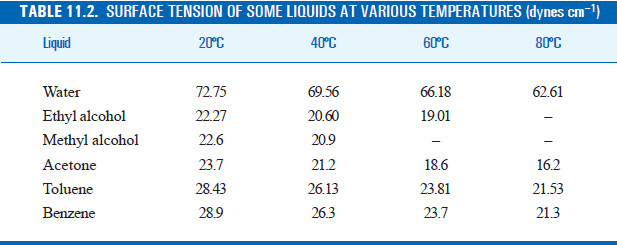
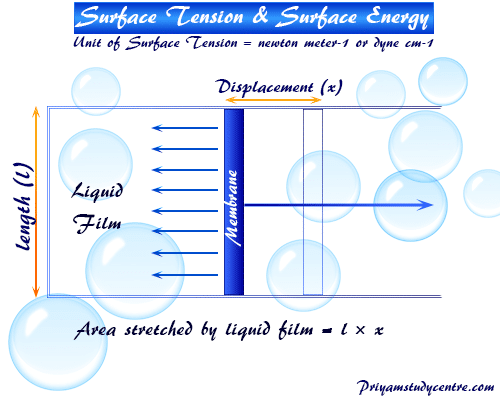
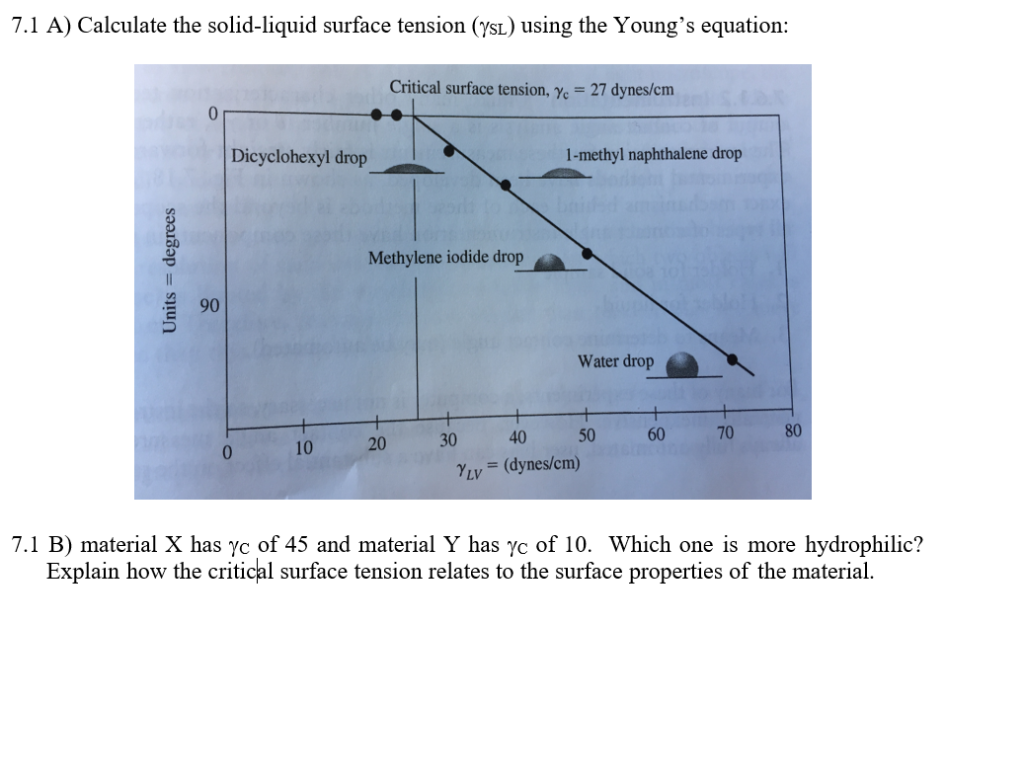
The surface tension of water at 20^∘C is 73 dynes cm^-1 . The minimum value of work done needed to increase surface area of water from 2 cm^2 to 5 cm^2 is

SOLVED: Table 1.6 Surface Tensions Liquid dynes /cm Acetone Benzene Ethanol Glycerol Mercury Methanol n-Octane Water 23.70 28.85 22.75 63.40 435.5 22.61 21.80 72.75

The surface tension of water at 0^0C is 75.5dyne/cm . Find surface tension of water at 25^0C ( alpha for water = 0.0021/K )

The surface tension of water is `75(dyne)/(cm)`. Find the minimum vertical force required to pull a - YouTube
Molecules | Free Full-Text | Calculation of the Surface Tension of Ordinary Organic and Ionic Liquids by Means of a Generally Applicable Computer Algorithm Based on the Group-Additivity Method

SOLVED: The surface tension for Ethanol and Propanol is ............. and ................ dynes/cm by drop weight method. The average mass of 20 drops of Water is 1.24 g The average mass of