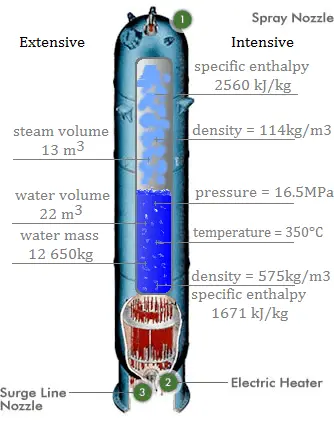
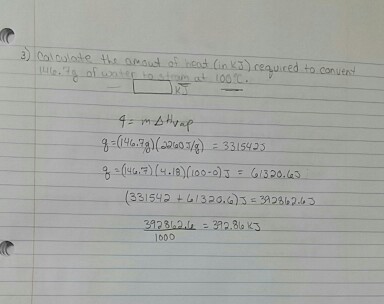SOLVED:What is the ΔH vap ^∘ of a liquid that has a vapor pressure of 621 torr at 85.2^∘ C and a boiling point of 95.6^∘ C at 1 atm ?

The enthalpy of vaporization of water at 100^o C is 40.63 KJ mol^-1 . The value Δ E for this process would be:

For a substance with the value of DeltaH(vap) and DeltaS(vap) given below, what is its normal boiling point in .^(@)C (DeltaH(vap)=59.0 kJ mol^(-1), DeltaS(vap)=93.65 Jmol^(-1))

SOLVED:For mercury, the enthalpy of vaporization is 58.51 kJ/mol and the entropy of vaporization is 92.92 J/K ? mol. What is the normal boiling point of mercury?

Standard enthalpy of vapourisation Delta("vap")H^(-) for water at 100^@C is 40.66 kJ mol^(-1). The internal energy of vapourisation of water at 100^@C (in kJ mol^(-1)) is (Assume water vapour to behave like an ideal gas).
Standard enthalpy of vaporisation ΔvapH° for water at 100°C is 40.66 kJ mol^-1. - Sarthaks eConnect | Largest Online Education Community

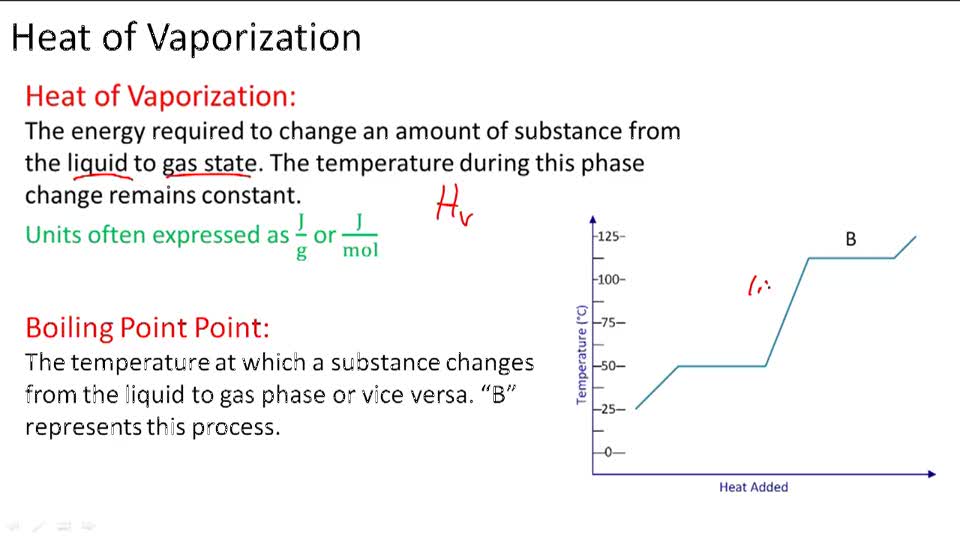
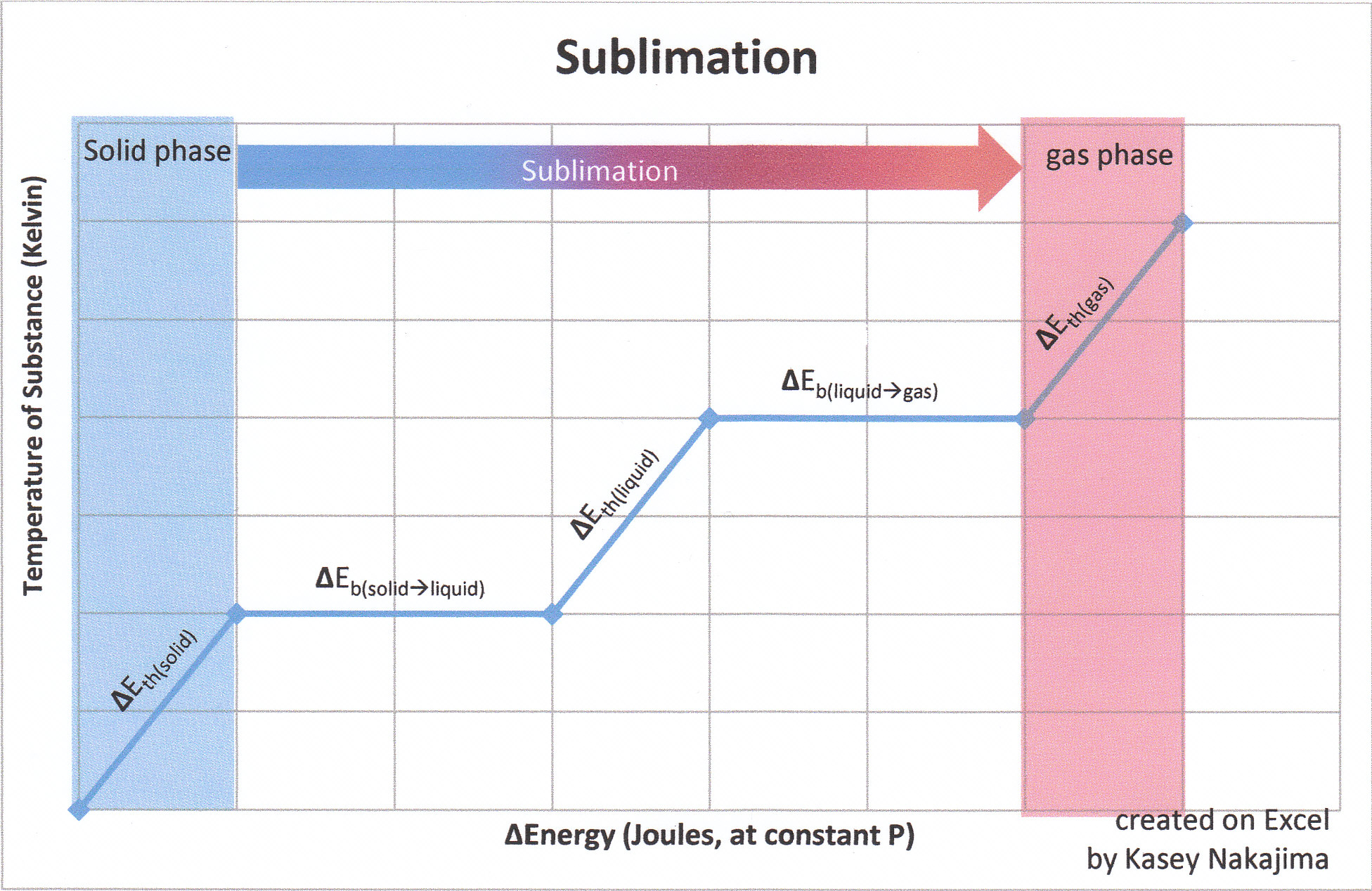
1 OBJECTIVES: –Classify, by type, the heat changes that occur during melting, freezing, boiling, and condensing. –Calculate heat changes that occur during. - ppt download




-438.png)