Intramolecular Electrostatic Effects on O2, CO2, and Acetate Binding to a Cationic Iron Porphyrin | Inorganic Chemistry

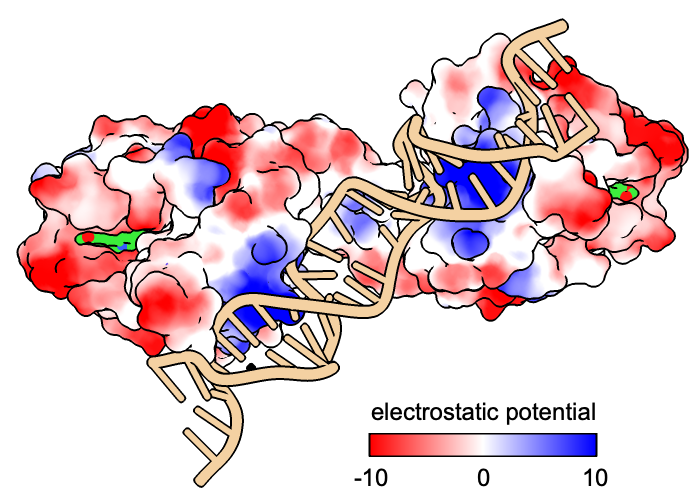
Electrostatic potential of the DNA binding surface of hApe1 and the corresponding surface area of Crc

Electrostatic Influence on the Kinetics of Ligand Binding to Acetylcholinesterase - Journal of Biological Chemistry

File:The electrostatic field lines of TRAP, trp RNA binding attenuation protein (PDB ID, 2EXS).jpg - Wikipedia

pH-Dependent Protonation of Histidine Residues Is Critical for Electrostatic Binding of Low-Density Lipoproteins to Human Coronary Arteries | Arteriosclerosis, Thrombosis, and Vascular Biology

Frustration in protein–DNA binding influences conformational switching and target search kinetics | PNAS

Electrostatic steering and ionic tethering in enzyme–ligand binding: Insights from simulations | PNAS

Tuning Electrostatic and Hydrophobic Surfaces of Aromatic Rings to Enhance Membrane Association and Cell Uptake of Peptides - Araujo - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Preferential binding of positive nanoparticles on cell membranes is due to electrostatic interactions: A too simplistic explanation that does not take into account the nanoparticle protein corona - ScienceDirect
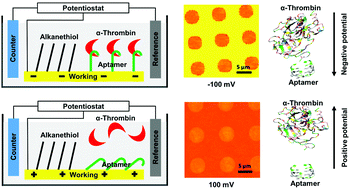
Aptamer based electrostatic-stimuli responsive surfaces for on-demand binding/unbinding of a specific ligand - Journal of Materials Chemistry B (RSC Publishing)

File:Molecular-Dynamics-Simulations-Suggest-that-Electrostatic -Funnel-Directs-Binding-of-Tamiflu-to-pcbi.1000939.s016.ogv - Wikimedia Commons

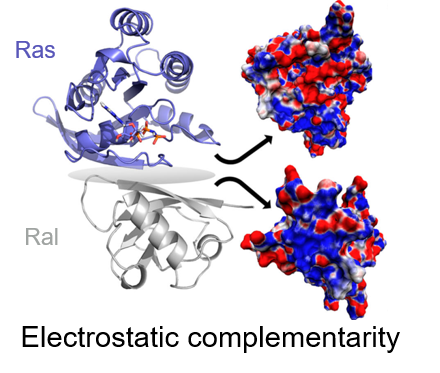
Electrostatic Complementarity as a Fast and Effective Tool to Optimize Binding and Selectivity of Protein–Ligand Complexes | Journal of Medicinal Chemistry
Molecular Dynamics Simulations Suggest that Electrostatic Funnel Directs Binding of Tamiflu to Influenza N1 Neuraminidases | PLOS Computational Biology

Electrostatic potential of the DNA binding surface of hApe1 and the corresponding surface area of Crc

SOLVED: What interactions may be most helpful when optimizing non-covalent binding of a competitive inhibitor to an enzyme? Specific electrostatic interactions near the active site Irreversible covalent bond formation: Binding to a