Thiophenol-Catalyzed Visible-Light Photoredox Decarboxylative Couplings of N-(Acetoxy)phthalimides | Organic Letters

Organocatalyzed Sulfa-Michael Addition of Thiophenols on Trisubstituted α-Fluoroacrylates, a Straightforward Access to Chiral Fluorinated Compounds | The Journal of Organic Chemistry

Recent Applications of Palladium‐Catalyzed Coupling Reactions in the Pharmaceutical, Agrochemical, and Fine Chemical Industries - Torborg - 2009 - Advanced Synthesis & Catalysis - Wiley Online Library
![Table 2 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing Table 2 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing](https://d3i71xaburhd42.cloudfront.net/24602ad7dd2ed0b911204ee266e0230c6219f654/3-Table2-1.png)
Table 2 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing
![Table 1 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing Table 1 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing](https://d3i71xaburhd42.cloudfront.net/24602ad7dd2ed0b911204ee266e0230c6219f654/2-Table1-1.png)
Table 1 from Palladium(0)-mediated rapid methylation and fluoromethylation on carbon frameworks by reacting methyl and fluoromethyl iodide with aryl and alkenyl boronic acid esters: useful for the synthesis of [11C]CH(3)--C- and [18F]FCH2--C-Containing
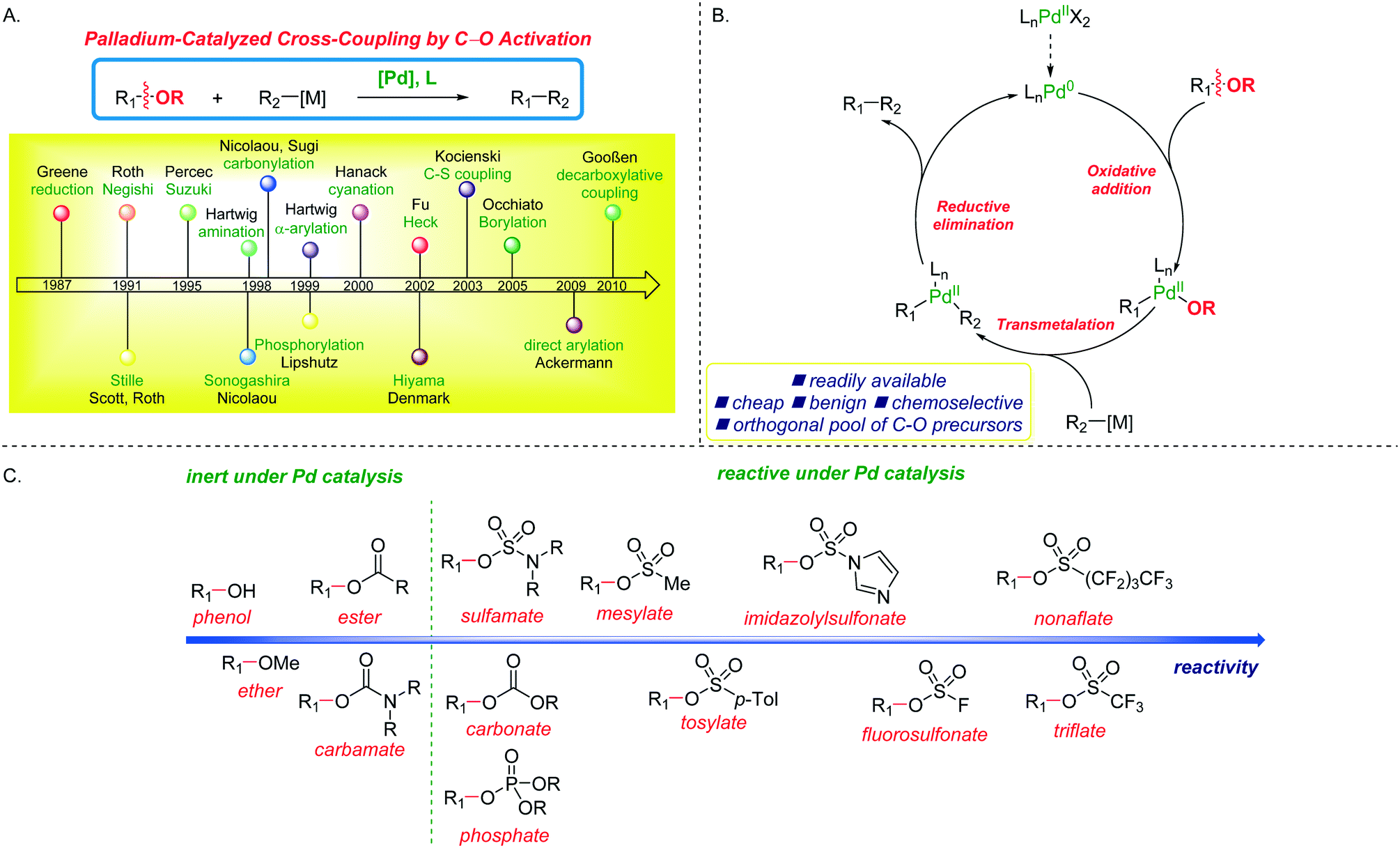
Palladium-catalyzed cross-couplings by C–O bond activation - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/D0CY01159B
1,2-DIFLUOROBENZENE (367-11-3) - Chemical Safety, Models, Suppliers, Regulation, and Patents - Chemchart

The Complementary Competitors: Palladium and Copper in C–N Cross-Coupling Reactions | Organometallics

Merging Electrolysis and Nickel Catalysis in Redox Neutral Cross-Coupling Reactions: Experiment and Computation for Electrochemically Induced C–P and C–Se Bonds Formation | CCS Chem

Anti-Markovnikov hydro(amino)alkylation of vinylarenes via photoredox catalysis | Nature Communications
![Tris(bicyclo[1.1.1]pentyl)phosphine: An Exceptionally Small Tri-tert-alkylphosphine and Its Bis-Ligated Pd(0) Complex | Journal of the American Chemical Society Tris(bicyclo[1.1.1]pentyl)phosphine: An Exceptionally Small Tri-tert-alkylphosphine and Its Bis-Ligated Pd(0) Complex | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/jacs.3c00885/asset/images/jacs.3c00885.social.jpeg_v03)
Tris(bicyclo[1.1.1]pentyl)phosphine: An Exceptionally Small Tri-tert-alkylphosphine and Its Bis-Ligated Pd(0) Complex | Journal of the American Chemical Society

Recent Applications of Palladium‐Catalyzed Coupling Reactions in the Pharmaceutical, Agrochemical, and Fine Chemical Industries - Torborg - 2009 - Advanced Synthesis & Catalysis - Wiley Online Library
![One-Pot Access to Benzo[a]carbazoles via Palladium(II)-Catalyzed Hetero- and Carboannulations | The Journal of Organic Chemistry One-Pot Access to Benzo[a]carbazoles via Palladium(II)-Catalyzed Hetero- and Carboannulations | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.6b02022/asset/images/acs.joc.6b02022.social.jpeg_v03)
One-Pot Access to Benzo[a]carbazoles via Palladium(II)-Catalyzed Hetero- and Carboannulations | The Journal of Organic Chemistry

Decarbonylative Fluoroalkylation at Palladium(II): From Fundamental Organometallic Studies to Catalysis | Journal of the American Chemical Society
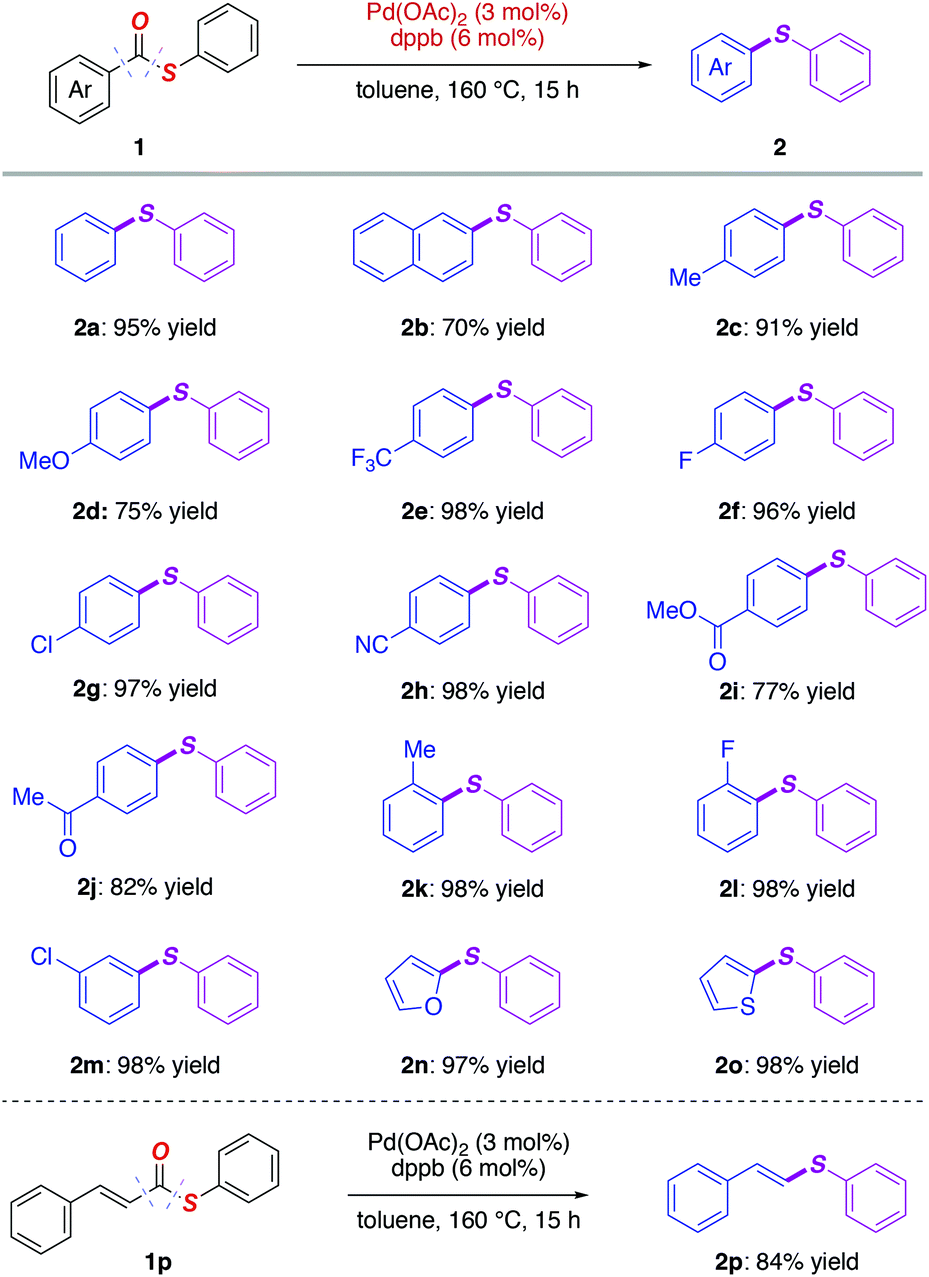
General and practical intramolecular decarbonylative coupling of thioesters via palladium catalysis - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO01576H

Regiodivergent Oxidative Cross-Coupling of Catechols with Persistent tert-Carbon Radicals | ACS Catalysis

Synthesis of 3-Iminoindol-2-amines and Cyclic Enaminones via Palladium-Catalyzed Isocyanide Insertion-Cyclization | The Journal of Organic Chemistry