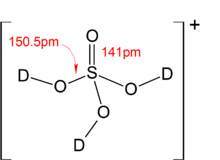
Sodium carbonate reacts with dil. H2SO4 to give the respective salt, water and carbon dioxide. - Sarthaks eConnect | Largest Online Education Community
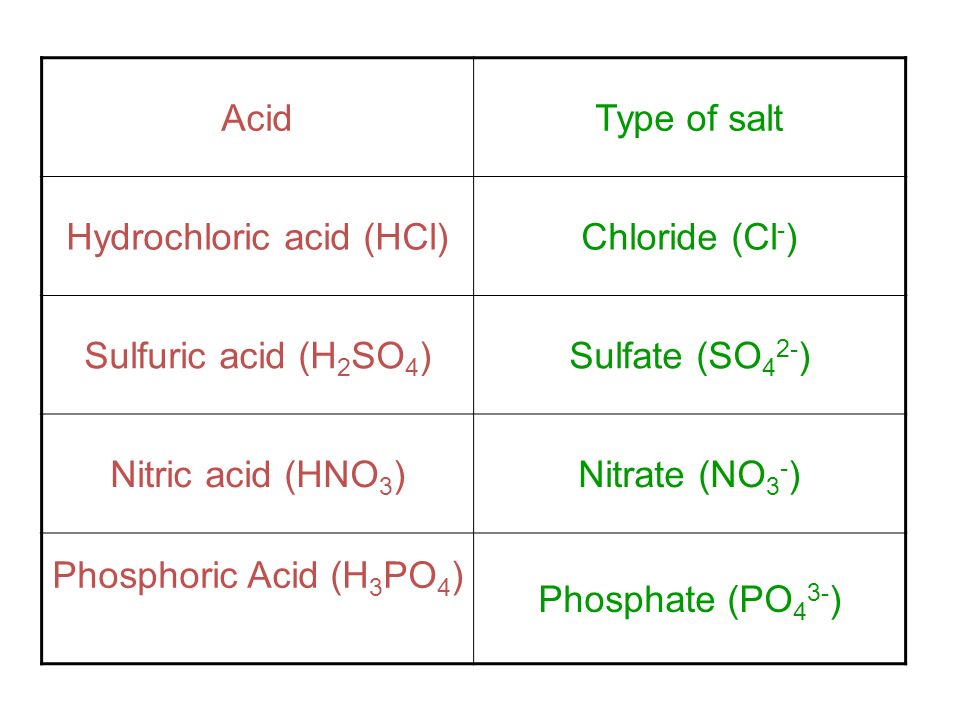
Sulphuric acid forms two types of salts with an alkali.Give reasons. - Sarthaks eConnect | Largest Online Education Community

A solution of a salt in concentrated H2SO4 turns a white paste of starch containing potassium iodide blue. The salt may be a:

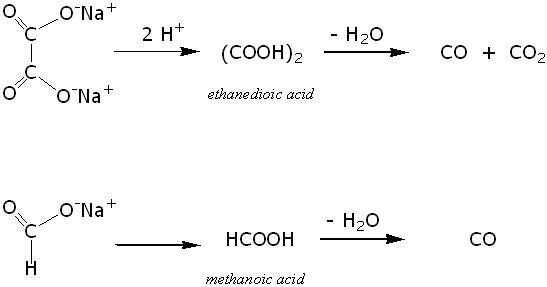
Sodium salt of an organic acid 'X' produces effervescence with conc.H2SO4 . 'X' reacts with the acidified aqueous CaCl2 solution to give a white precipitate which decolourizes acidic solution of KMnO4 'X'

Sulfuric Acid LO: Outline uses and reactions involving Sulfuric Acid Starter: What is an acid? - ppt download

Assertion: H2SO4 forms only one series of salts.Reasons: The molecules of H2SO4 consists of only one OH group.