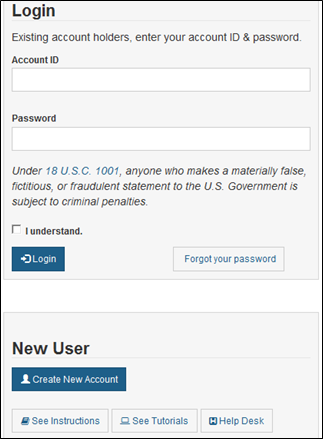
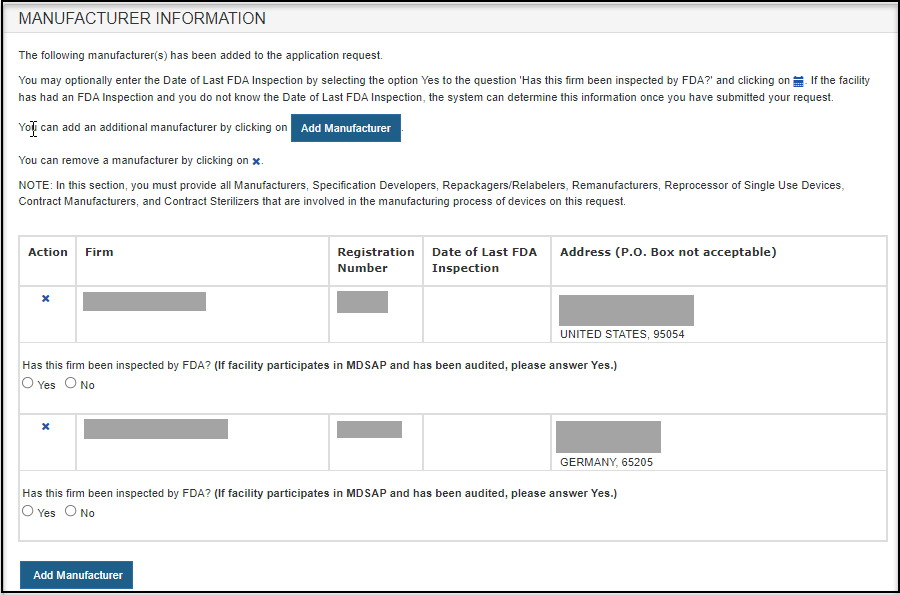
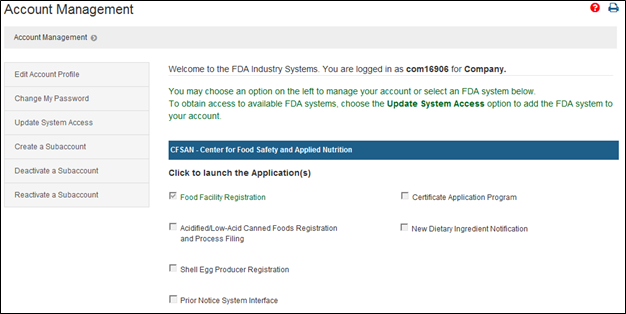
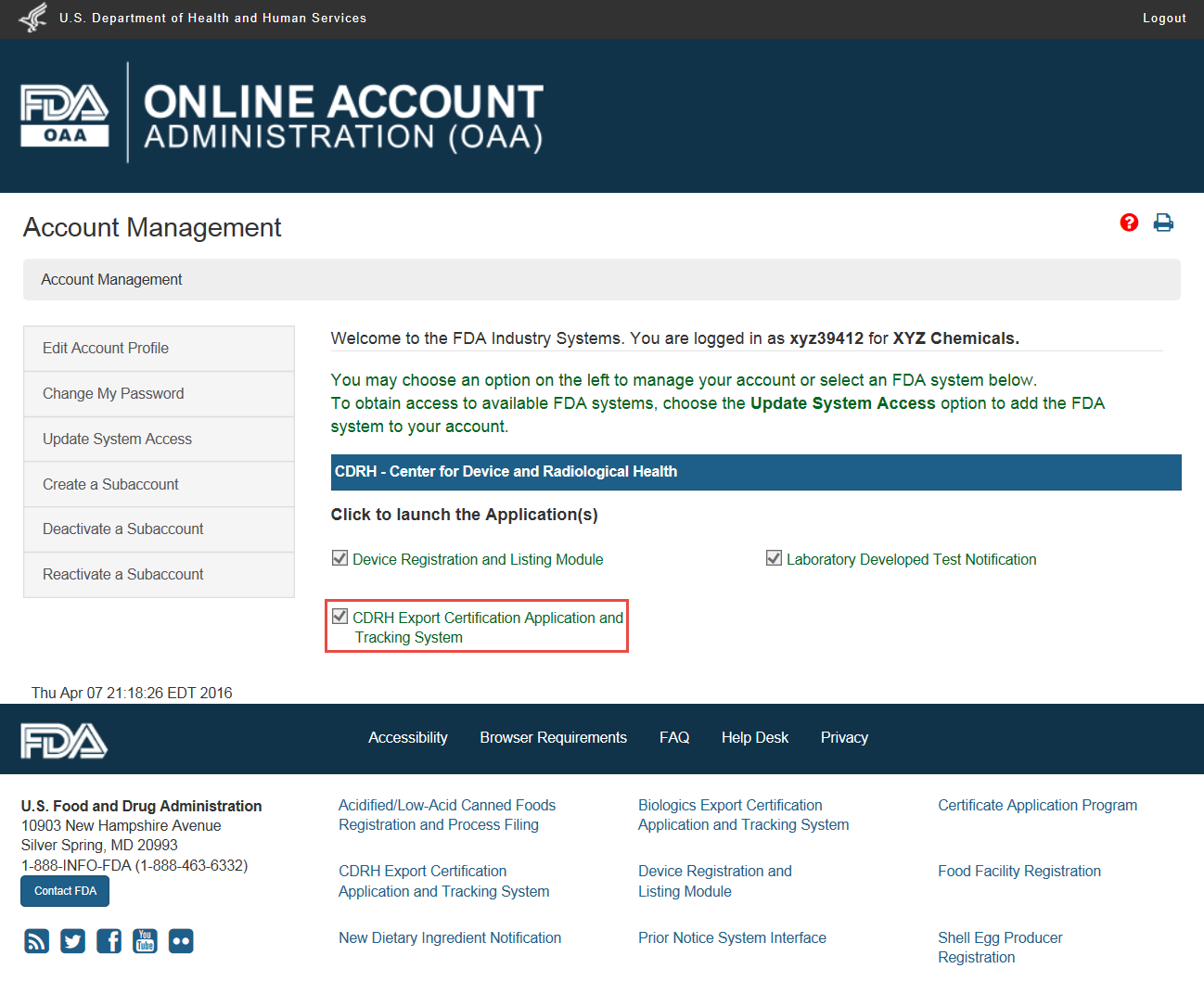
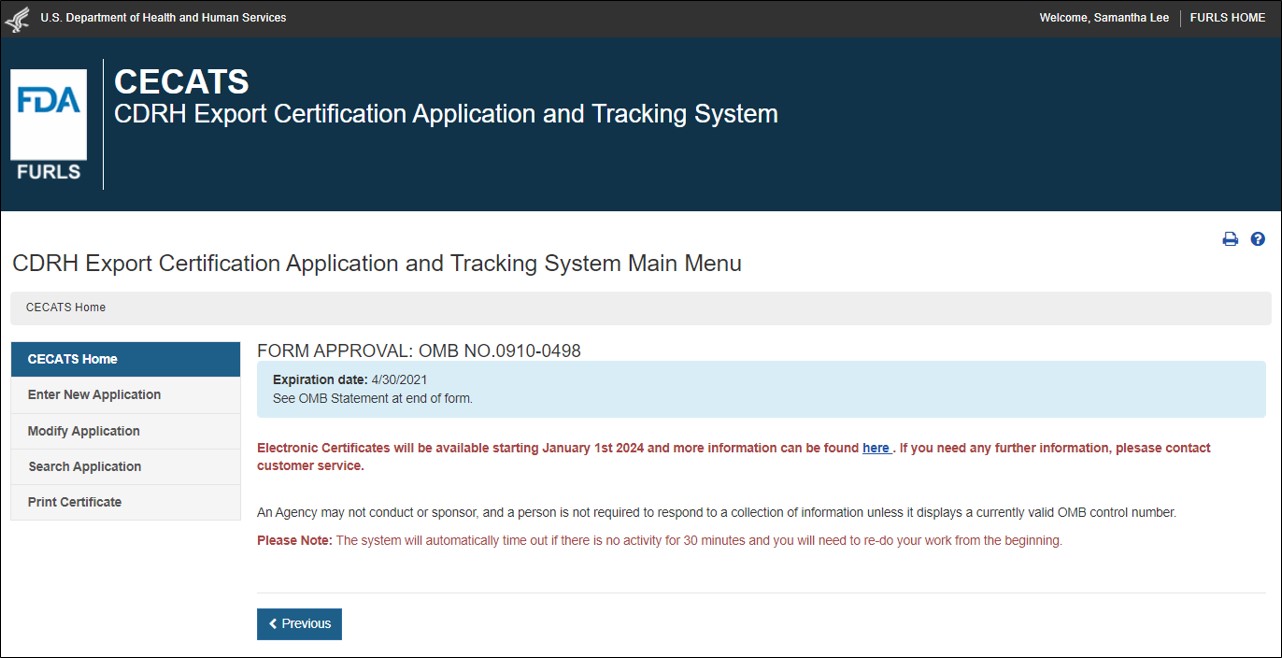
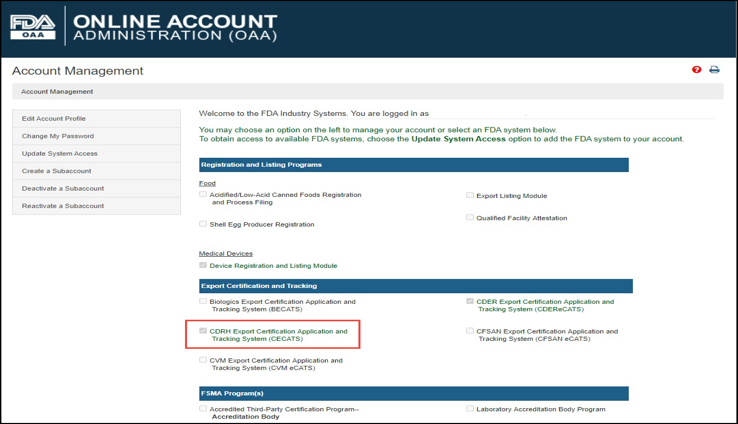
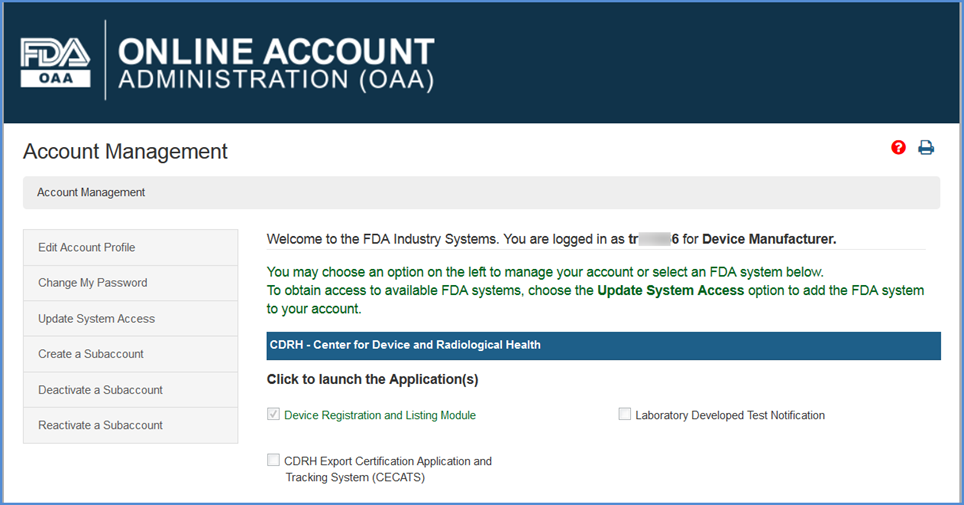
Foreign Supplier Verification Programs (FSVP) Importer Portal for FSVP Records Submission User Guide

Structural Alert/Reactive Metabolite Concept as Applied in Medicinal Chemistry to Mitigate the Risk of Idiosyncratic Drug Toxicity: A Perspective Based on the Critical Examination of Trends in the Top 200 Drugs Marketed

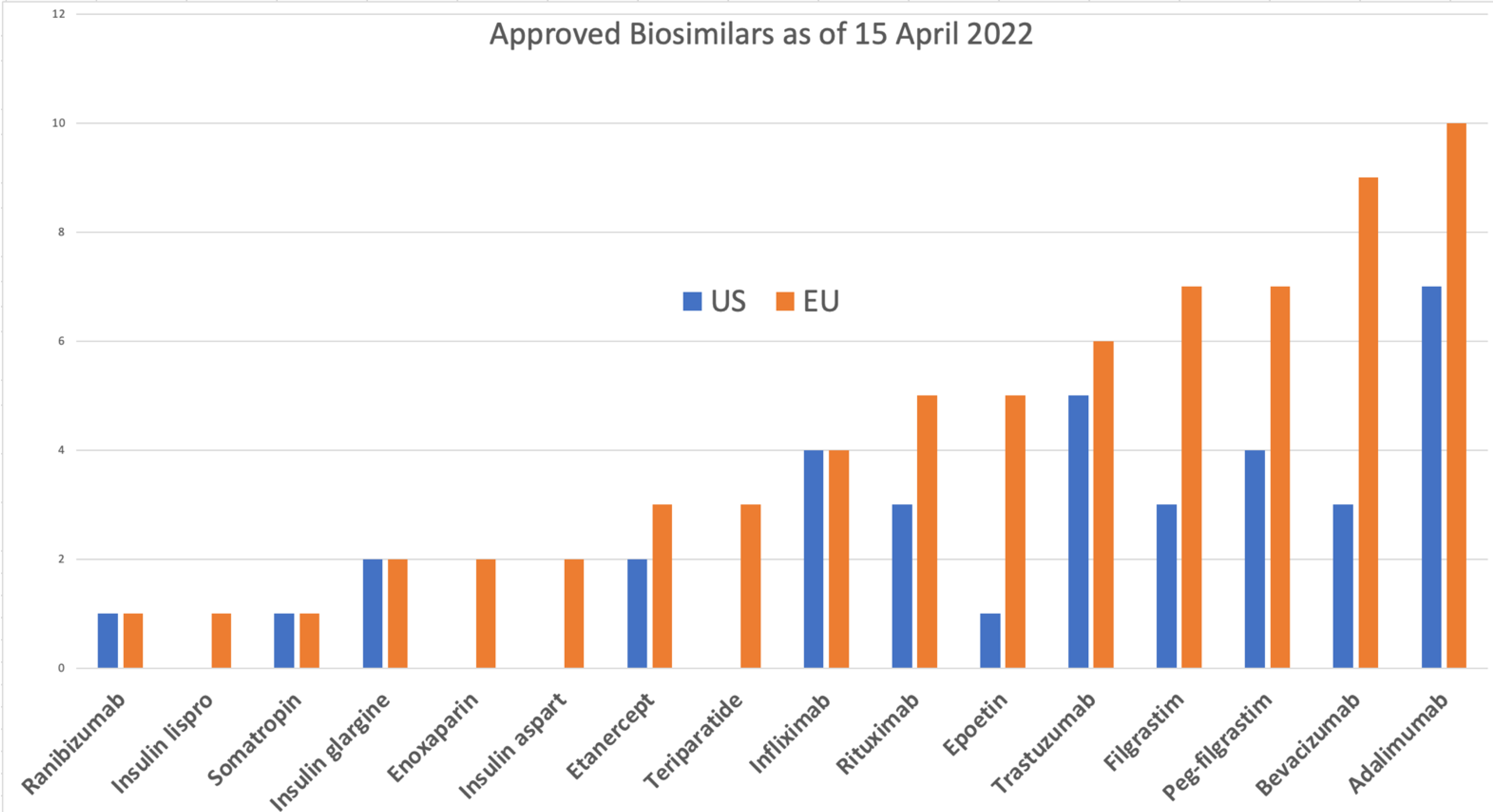
FDA's Interpretation Of The “Deemed To Be A License” Provision Of The Biologics Price Competition An | Contract Pharma

Meeting FDA Guidance recommendations for replication-competent virus and insertional oncogenesis testing: Molecular Therapy - Methods & Clinical Development
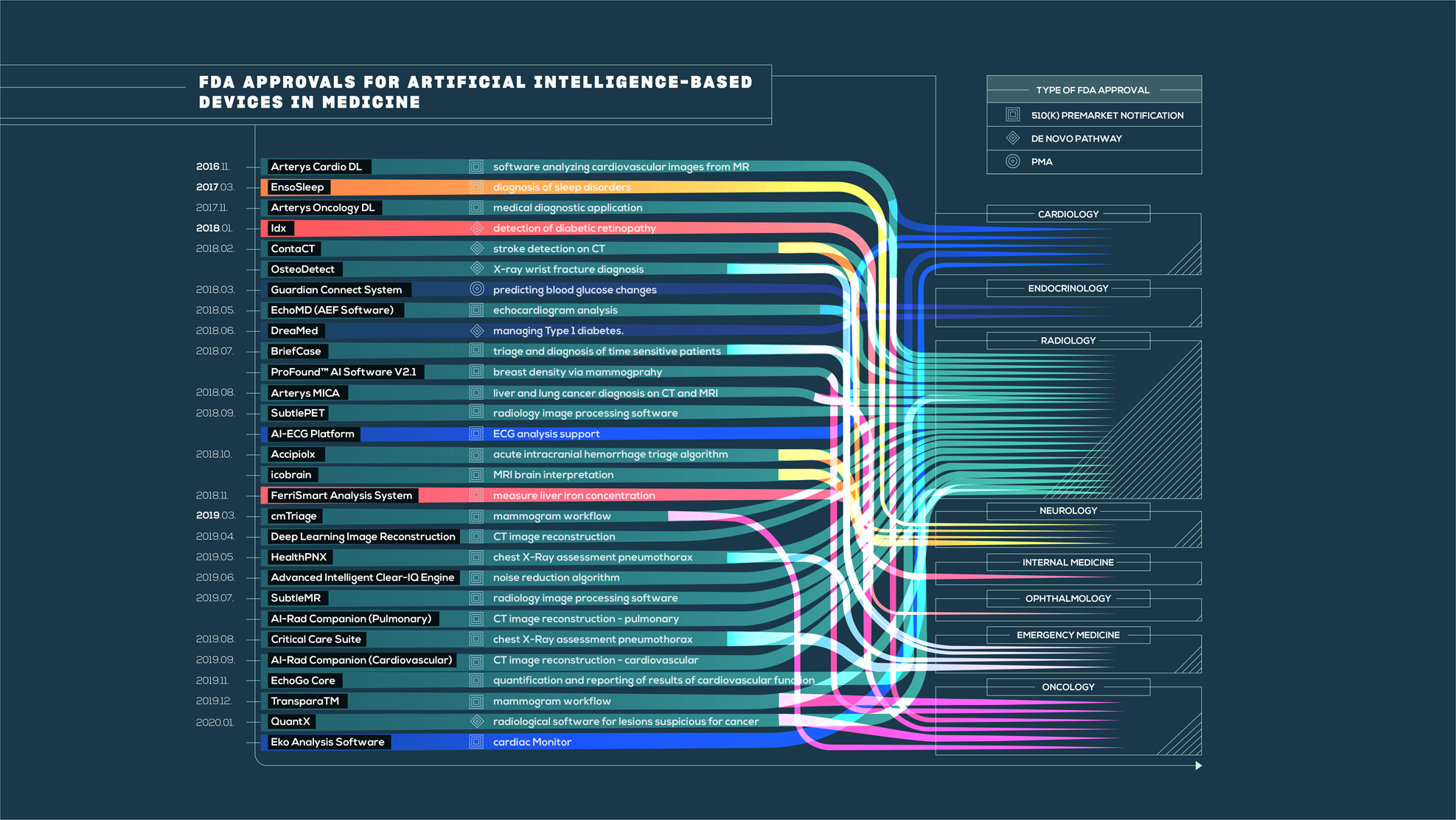
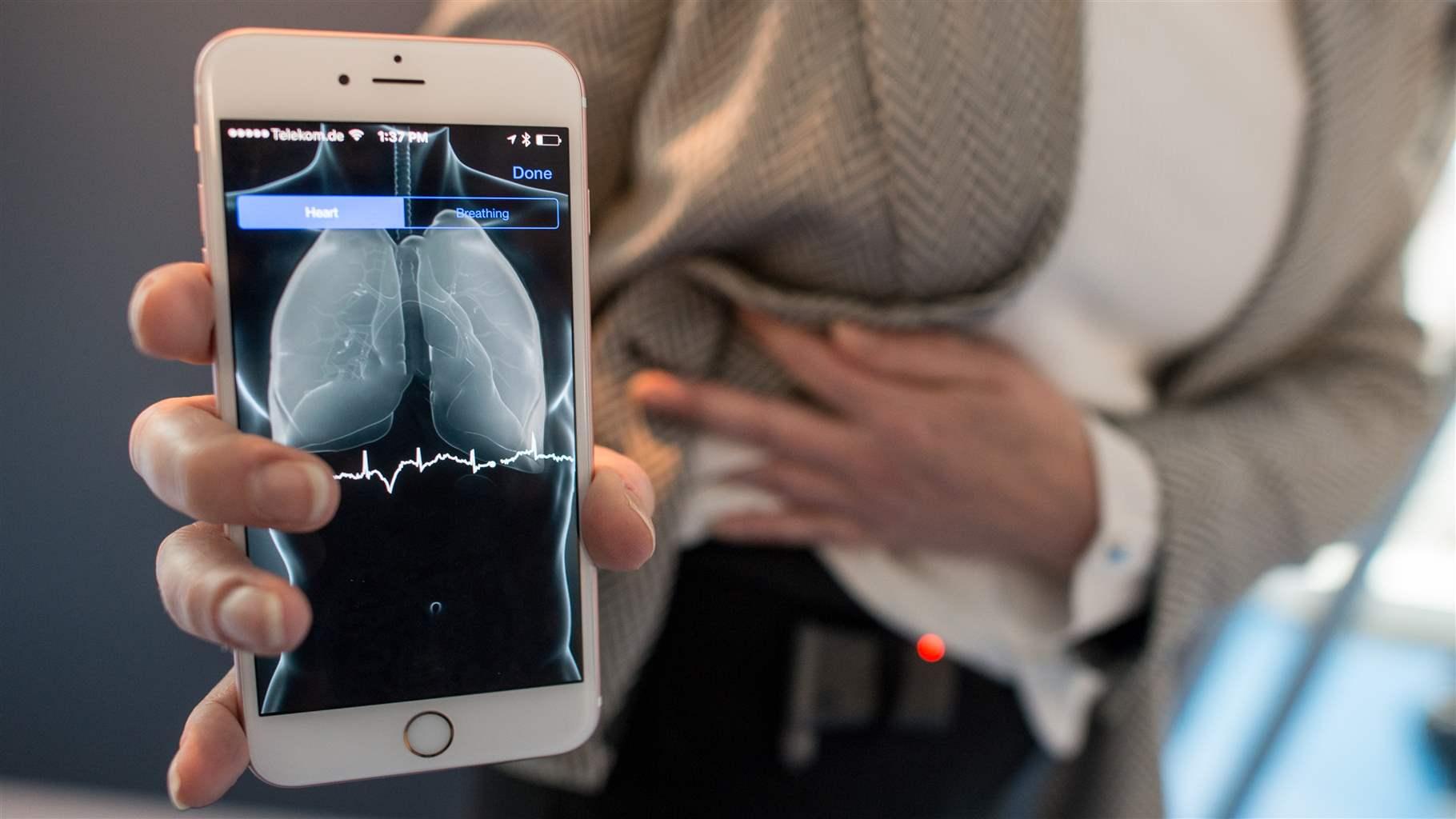
The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database | npj Digital Medicine
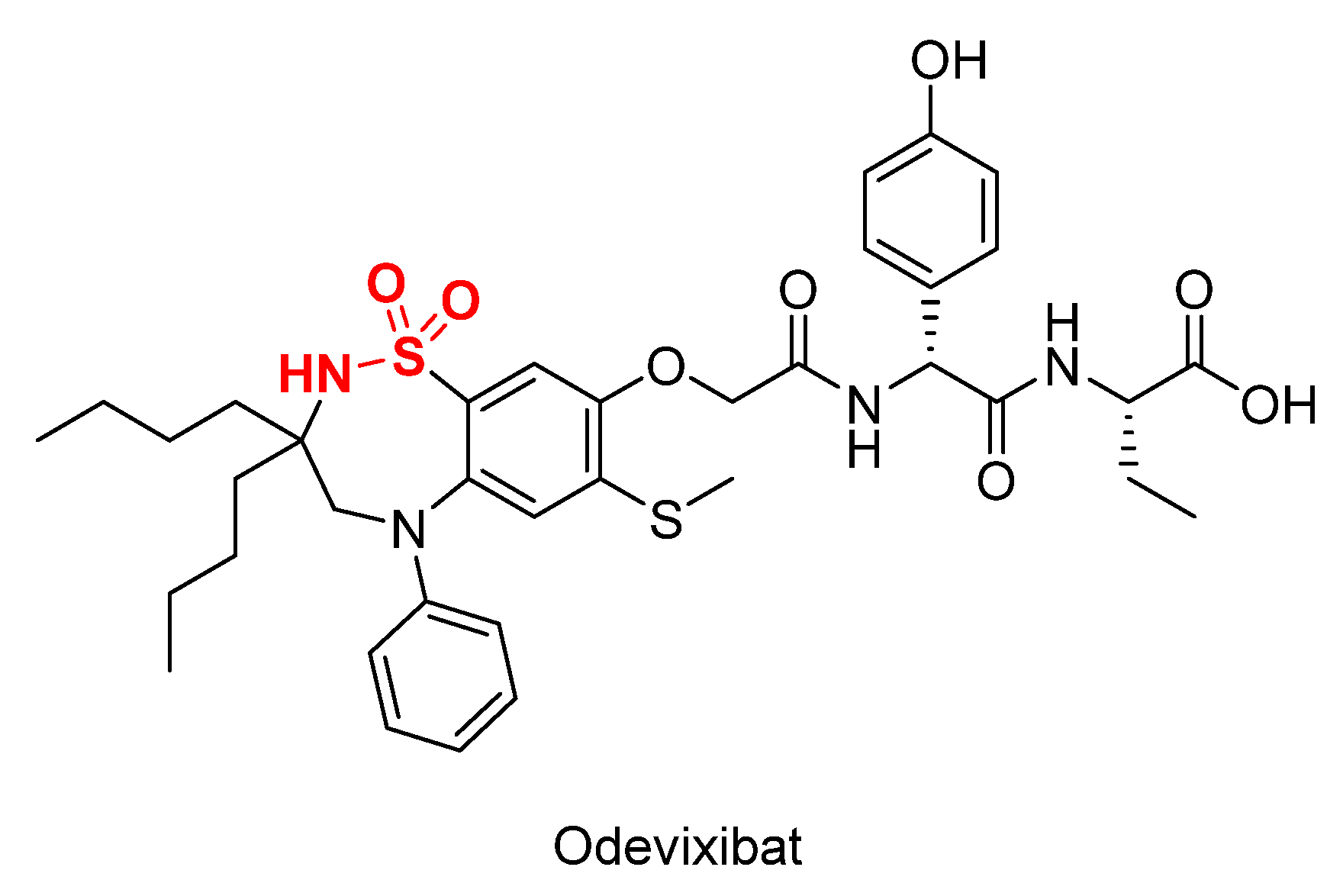
Pharmaceutics | Free Full-Text | FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis
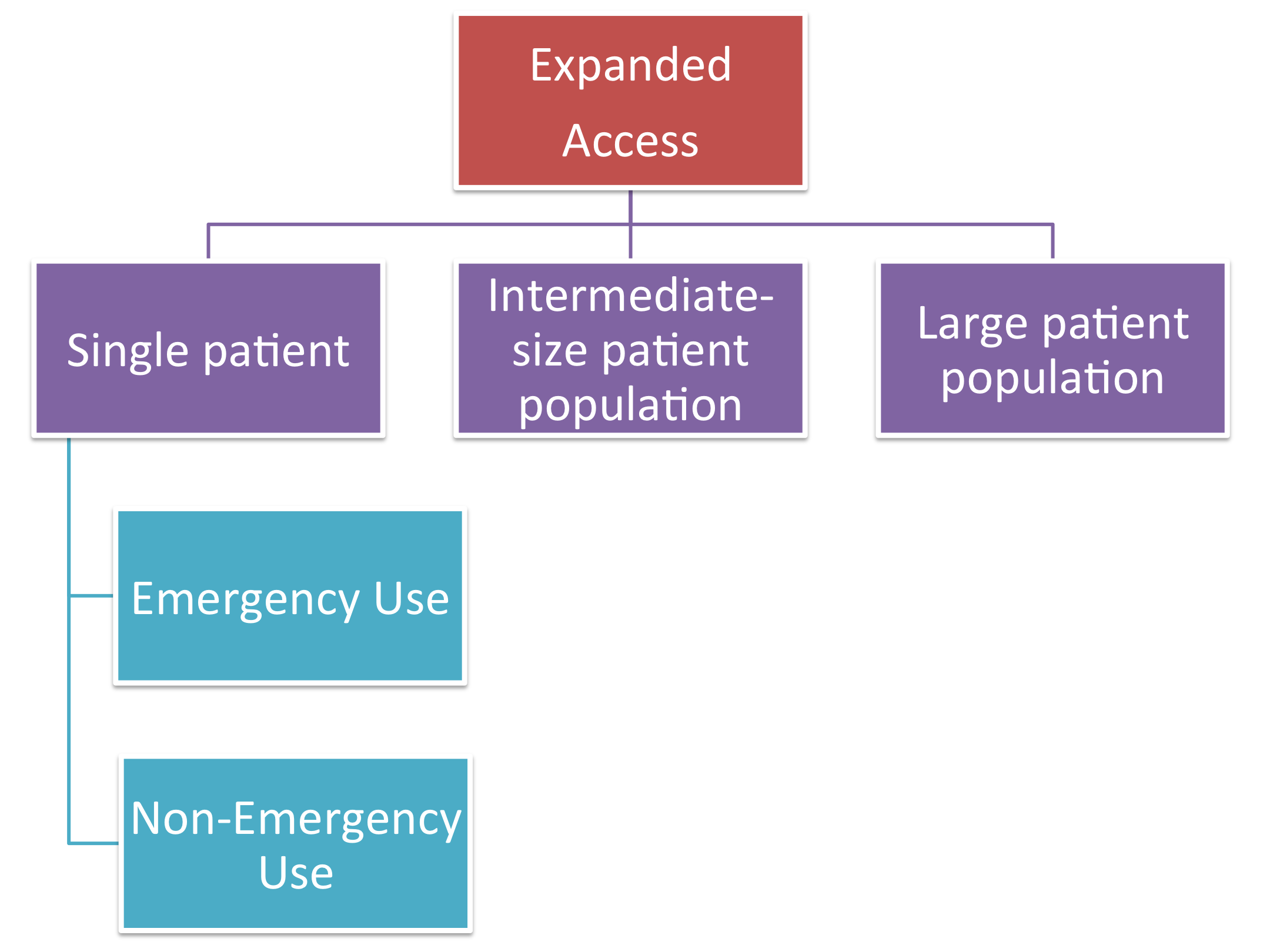
FDA Emergency Use Authorization: A Brief History From 9/11 to COVID-19 - Food and Drug Law Institute (FDLI)

Summary of Guidance for Minimizing the Impact of COVID-19 on Individual Persons, Communities, and Health Care Systems — United States, August 2022 | MMWR

The FDA Verification Portal 🔎 Check if an establishment is licensed and/or verify if a health product is registered with the Food and Drug... | By Food and Drug Administration Philippines