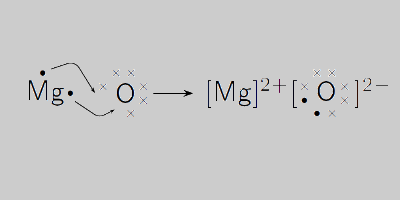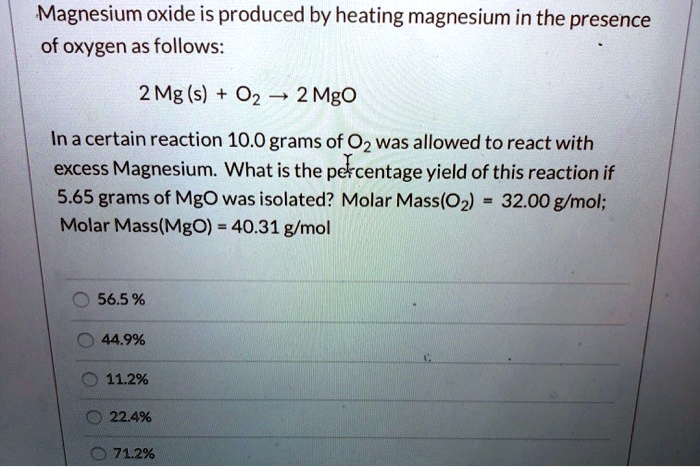
SOLVED: Magnesium oxide is produced by heating magnesium in the presence of oxygen as follows: 2 Mg (s) 02 2 Mgo In a certain reaction 10.0 grams of 0z was allowed to

OB: Intro to the Mole You gotta have a reference table and a calculator of your own now. No exceptions. - ppt download
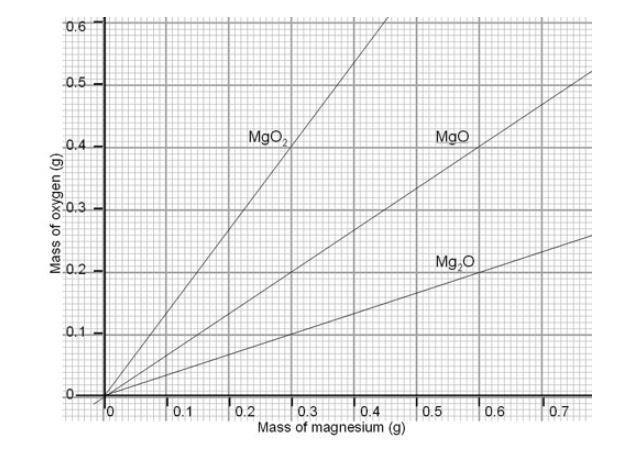
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as

1a.calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 - Brainly.com

N. Calculate the number of moles ofmagnesium oxide, MgO in i. 80 g andii. 10 g of the compound. - Brainly.in
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/9085118/105391b5-2617-461a-9970-1a42367b289a.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]

Question Video: Calculating the Mass of Oxygen Required to React with a Given Number of Magnesium Atoms | Nagwa
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/9085118/c1781a1a-7e5f-47e8-a9bf-9b6d725abef3.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]

PDF) Experimentally Determining of the Empirical Formula of Magnesium Oxide | Folk Narongrit - Academia.edu
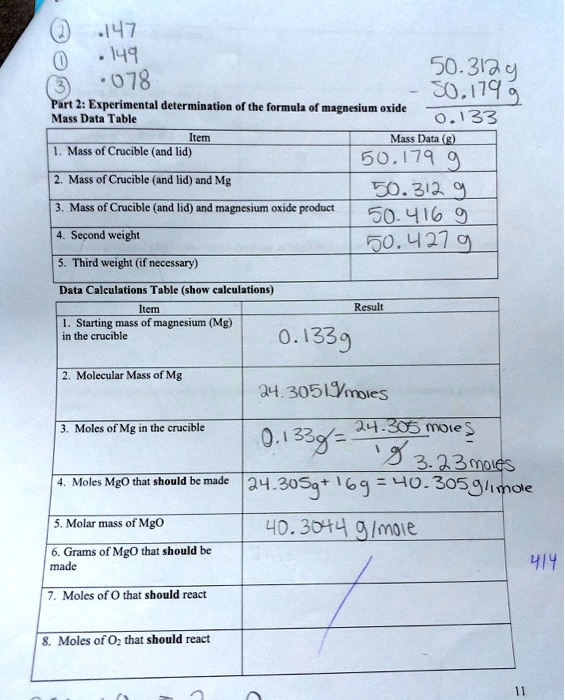
SOLVED: .147 149 50.313,9 078 50.179 Part 2: Experimental determination of the formulz of magnesium oxide Mass Data Table 0.133 Ms? Data Mass of Cricible (and lid) 50.179 Mass of Crucible (and